| Issue |
Int. J. Lim.
Volume 59, 2023
|
|
|---|---|---|
| Article Number | 3 | |
| Number of page(s) | 14 | |
| DOI | https://doi.org/10.1051/limn/2023002 | |
| Published online | 27 February 2023 | |
Research Article
Zooplankton egg bank: characterization and effect of biotic factors on hatching
1
Posgrado en Ciencias del Mar y Limnología, Universidad Nacional Autónoma de México, Circuito exterior, Cd. Universitaria, Coyoacán 04510, Ciudad de México, Mexico
2
Laboratorio de Producción Acuícola, Universidad Nacional Autónoma de México, Campus Iztacala, CP 54090, Los Reyes Iztacala, Tlalnepantla, Estado de México, Mexico
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
1
July
2022
Accepted:
6
January
2023
Abstract
Many zooplankton organisms facing harsh environmental conditions producing resting eggs. Resting eggs accumulate in the sediment and create a resting egg bank. Knowledge on the egg bank structure, morphology and morphometry of the eggs as well as the effect of cues on hatching, contributes to the understanding zooplankton dynamics in lakes. Here we described the composition and structure of zooplankton egg bank from a shallow tropical lake and analyzed the effect of some biotic factors on hatching rates. In order to describe the structure and composition of the resting egg bank, we determined the richness, density, morphology and morphometry of the resting eggs isolated by the sugar flotation method. Diapausing eggs were measured and their external features studied by microphotography under optical microscope and SEM. To analyze the effect of biotic factors on hatching rates, we exposed the resting structures and the entire sediment to three biotic factors: a chlorophyte (Scenedesmus acutus), a cyanobacteria (Microcystis sp.) and a predatory rotifer (Asplanchna girodi). A total of 25 zooplankton species hatched from the sediments. Our results show that the medium density of the healthy-looking diapausing eggs was of 7.6 ± 2 diapausing eggs · cm−3 and that rotifers are the predominant group in the egg bank. Medium conditioned with the chlorophyte and the cyanobacteria resulted in a higher hatching rate. The ornamentations of rotifer diapausing eggs present different features, such as wrinkles and spines, which can help to taxonomic identification without the need of promoting hatching.
Key words: Diapausing eggs / density / morphology / morphometry / rotifers
© EDP Sciences, 2023
1 Introduction
Dormancy is an inactive stage of several invertebrates which normally follows sexual reproduction and is considered a strategy to survive fluctuating environmental conditions (Alekseev et al., 2007; Stelzer and Lehtonen, 2016). This mechanism has been observed in different groups of invertebrates, both terrestrial and aquatic (Gill et al., 2017). A clear example of aquatic organisms that conserve dormancy, particularly diapause, in their life cycles are the three principal components of freshwater zooplankton, rotifers, cladocerans, and copepods (Hairston and Fox, 2009).
Rotifers and cladocerans combined sexual and asexual reproduction in their life cycle (Dodson et al., 2009; Decaestecker et al., 2009; Fontaneto and De Smet, 2015). The asexual phase predominates, while the sexual phase, finally resulting in the production of diapausing eggs (DE's), is often induced by environmental conditions such as the photoperiod (Gilbert, 1977), temperature (Kogane et al., 1997; Gyllström and Hansson, 2004), availability and quality of the food (Kleiven et al., 1992; Gilbert, 2010) and intrinsic factors (Gilbert, 1974; Lass et al., 2005), of which the age and genotype of females (Pourriot and Clément, 1977) and population density are important (King and Snell, 1980; Gilbert, 2003). In copepods, diapause involves resting stages (not developed embryos) or, in some cases, encysting of the larval phase (Marcus, 2005). Diapause is observed mainly in calanoids, because the other taxonomic groups present quiescence as latency strategy (Marcus, 2005). Calanoid copepods maintain a constant sexual reproduction; however, during specific environmental stimuli they are capable of changing the type of egg that will be produced, subitaneous or DE's (Zeller et al., 2004). In the aforementioned groups induction of diapause is considered as the result of adverse environmental conditions (Gyllström and Hansson, 2004). Recent studies, however, indicate that mictic females exist at all times in populations to ensure sexual reproduction and DE's that can withstand harsh conditions (Gilbert, 2007).
Once produced, the DE's sink through the water column until they reach the sediment, forming what is known as the egg bank (Hairston and Fox, 2009). The egg bank is where the resting eggs produced in different periods accumulate and new genetic variants are recruited for the active populations (Brendonck and De Meester, 2003; De Stasio, 2007). According to the literature, resting stages occur at very variable densities in the egg bank of most aquatic ecosystems (Brendonck and De Meester, 2003; Walsh et al., 2017). Particularly in wetlands, the resting stages have a greater richness and density (Snell, 1983; De Stasio, 2007; García-Roger, 2008). However, few studies evaluate the temporal and spatial variations of the DE's (Duggan, 2002; Maia-Barbosa et al., 2003; García-Roger et al., 2006), so our understanding of their importance in dynamic populations of zooplankters is still limited.
Typically, in rotifers and cladocerans, taxonomic importance is attributed to the DE's, since their ornamentations have a species-specific character (Gilbert and Wurdak, 1978; Vandekerkhove et al., 2004). However, according to Walsh et al. (2017), there is currently no direct evidence of the presence of DE's in about 80% of the rotifer species; therefore, their morphological and morphometric knowledge is scarce. Recently, Guerrero-Jiménez et al. (2019) observed substantial evidence of the species-specific character of 13 rotifers DE's collected in Mexico and Spain, using light microscopy and scanning electron microscopy. These results confirm that these microscopy tools are a valid alternative for the identification of DE's, compared to other strategies, such as molecular methods, considered the best in the identification of DE's (Moreno et al., 2017). Studies that clarify the taxonomy of DE's would enable their use as a complementary tool in ecological work (May, 1986; Vandekerkhove et al., 2004: 2005b).
On the other hand, the DE's have a great biogeographic, evolutionary and ecological importance for zooplankton, since they facilitate their dispersion (Moreno et al., 2016), increase the genetic variability (Brendonck and De Meester, 2003) and allow evasion of adverse environmental conditions (e.g. drying, low temperatures, anoxia, predation, food shortage) for long periods (Gyllström and Hansson, 2004). According to this, the DE's would hatch when the conditions of a water body are reestablished or as soon as they are in adequate conditions (Hairston et al., 2000). However, the literature has focused on the mechanisms related to the induction of the DE's, while the mechanisms of diapause termination concerning the conditions, times, and factors involved in hatching are meager (Taylor, 1980; De Stasio, 2007; Walsh et al., 2014). Both, the induction of DE's and their hatching, are processes that favor the permanence of the species in the systems and should be related to one or more signals to avoid delays (Gyllström and Hansson, 2004). Any delay in breaking diapause and the beginning of the hatching of the DE's, would leave the individuals in unfavorable conditions, so the timing and signals associated with hatching must be under strong selection pressure (Ślusarczyk and Flis, 2019).
Therefore, the resumption of the life cycle in rotifers and cladocerans from the DE's to the direct development, seems to be exclusively associated with abiotic cues such as photoperiod, temperature and dissolved oxygen concentration (Pourriot and Snell, 1983; Ricci, 2001; Vanderkerkhove et al., 2005a). Although, in some cases, spontaneous hatching of DE's has been recorded (Gilbert and Schröder, 2004) and in other cases the presence of light regardless of the photoperiod (Ślusarczyk and Flis, 2019), desiccation of the sediments (Vargas et al., 2019), salinity (Conde-Porcuna et al., 2018) or even artificial products have been successful as hatching inducers (Balompapueng et al., 1997; Kanagasabapathi and Munuswamy, 2011). Likewise, there is evidence that some biotic signals have an effect on the termination of diapause in zooplankton, mainly in cladocerans (Bozelli et al., 2008). The exposition of the DE's of Daphnia magna with fish kairomones, showed an increase in the hatching rate (Lass et al., 2005), while DE's of Daphnia obtusa incubated with kairomones of the fish Rutilus rutilus decreased the hatches success (Bozelli et al., 2008). In the case of the cladocerans, Moina micrura and Diaphanosoma birgei, the presence of kairomones had no effect on the hatching rate of their DE's (Santangelo et al., 2010). In these works, the responses to the kairomones were dissimilar. In rotifers, a positive effect on hatching of DE's has been observed when incubated with live algae (Minkoff et al., 1983), additionally, it has been observed that the exposure of DE's to kairomones of the predatory rotifer Asplanchna is related to faster or higher hatching (Yin et al., 2021). Finally, in copepods, contradictory relationships have been described between phytoplankton, particularly diatoms, and the success in the hatching of eggs. For some authors, a deleterious effect exists (Miralto et al., 1999; Paffenhöfer et al., 2002) and for others, there is no such negative effect (Irigoien et al., 2002). Therefore, the hatching of the DE's could be related to both types of biotic and abiotic signals separately or even work in combination. However, the insipient number of studies on biotic signals in the literature and the relative ease of experimenting with abiotic factors can explain this trend (Koštál, 2006).
In natural populations, the hatching of the resting stages could be related to biotic factors, for example changes in limiting factors (Gyllström and Hansson, 2004; Koštál, 2006). In tropical systems environmental factors such as temperature and photoperiod show a not so marked variation throughout the year (Lampert and Sommer, 2007), the hatching of the resting stages could be stimulated by other signals, such as the concentration of food in the medium or any other allelochemical-mediated signal (Koštál, 2006). Particularly in tropical countries where cyanobacterial blooms are common and potentially harmful (Mowe et al., 2015), they could be associated as a signal of hatching, because the blooms, for example of Microcystis, have been related to a specific season of the year (dry season) or even because of their potential toxicity (Mowe et al., 2015), could negatively affect the egg bank. In this study, we describe the composition of the resistance egg bank through species richness, abundance, morphology, and morphometry of DE's. Additionally, we used two approaches to evaluate the effect of three biotic factors, two potential resources (Scenedesmus acutus and Microcystis sp.) and a predator (Asplanchna girodi), on hatching DE's patterns. First using the DE's isolated via hatching rates and the second with the entire sediments through hatching. We hypothesized that DE's could detect these biotic signals by modifying their species-specific response and hatching patterns.
2 Methods
2.1 Study area
Tilostoc wetland, is located in central Mexico 19° 13' 00”N; 100° 11' 00”W in Valle de Bravo, State of Mexico (Fig. 1). It is a shallow (between 1.5 and 1.70 m), eutrophic and high altitude (1800 m a.s.l) water body connected to the Valle de Bravo reservoir, which belongs to the Cutzamala hydraulic system, and contributes one third of the water supplied to Mexico City's Metropolitan Area (Ramírez-Zierold et al., 2010).
 |
Fig. 1 Sample points in Tilostoc wetland (MZ = Macrophyte Zone, PZ = Pelagic Zone, and LZ = Littoral Zone) and its location in Mexico State, Mexico. The grey area represents the part of waterbody with presence of macrophytes of genre Tipha. |
2.2 Sediment samples
The sediment samples were collected on May 2017 only in the east part of the Tilostoc wetland, since the rest of the water body is covered by macrophytes of the genera Typha (Fig. 1). Considering the heterogenic distribution of wetland egg banks (Snell et al., 1983; Brock et al., 2005), we sampled in three different zones in order to increase species richness and to reduce the variance within the wetland. However, we did not perform mixed composite samples (pooled samples) and presented our results for the three sites separately but without comparing them. At each site, we collected 1100 cm3 of sediment, with an Ekman grab (15 × 15 cm). We only considered the most recent stratum; therefore we carefully removed the first 5 cm of sediment from the top of the grab (Duggan et al., 2002; Bhusnale et al., 2016) and transferred them to resealable plastic bags. The sediments were transported to the laboratory and maintained in dark conditions to inhibit the hatching before the experiments (Santangelo et al., 2011) and at 4 °C, conditions that promote higher hatchings rates in rotifers (Chittapum et al., 2015). The DE's were not incubated in filtered lake water since it can contain several signals that potentially cause interference in experimental results.
2.3 Species richness
We took 18 cm3 of sediment (20 g wet weight) previously homogenized and placed it in a Petri dish with 30 mL of distilled water for observation in a stereoscopic microscope. The DE's were isolated and classified considering their morphological features, which were photographed, and some DE's of each morphotype were placed individually in a 24 well cell culture plate of 3.5 mL capacity with 1.5 mL of distilled water (Portinho et al., 2018) at a 25 °C temperature and with constant light to promote hatching (Albritton and White, 2006). When the DE's hatched, the organisms were isolated and identified using specialized taxonomic keys. For rotifers we used keys of Koste (1978) and Shield (1995), and when the trophi was necessary we extracted it from the individuals by dissolving them with sodium hypochlorite (commercial grade). For cladocerans and copepods (culture after hatching), we observed the different appendages of individuals to reach the lowest possible taxonomic level using the keys of Elías-Gutiérrez et al. (2008). Additionally, we used the hatching experiment, which will be described ahead, to complement total species richness, due to some resting stages were not observed with the isolate through stereoscope microscope, but they appeared in direct hatching from sediment.
2.4 Resting stages density
We used the method of isolation of Onbé (1978) for which we diluted 4.5 cm3 (5 g wet weight) of sediment in 40 mL of a 1:1 solution of distilled water and sucrose. The solution was stirred during 5 min and immediately after centrifuged at 2500 rpm for 5 min. The supernatant was filtered through a 20 µm mesh, and finally washed with distilled water until the sucrose was completely removed. The isolated material was placed in a Petri dish and observed in a stereoscopic microscope where resting stages were counted. We determined the density in the sediments only by taking into consideration the healthy-looking DE's, that were determined according to the criteria established by García-Roger et al. (2006) for rotifers and, for cladocerans we used the criteria of Brandão et al. (2014) and Conde-Porcuna et al. (2014). We made four replicates at each sampling site to obtain the density of resting stages (expressed in resting stages cm−3).
2.5 Morphologycal analysis
The morphology of resting stages was performed by observing scanning electron microscopy (SEM) according to the criteria of De Smet (1998). The DE's isolated by the technique of Onbé (1978) were submitted to a series of washes with ultrapure water, then placed on aluminum plates where they were dehydrated during 24 h and finally coated with a layer of colloidal gold. Then, the DE's were observed in an electronic microscope Jeol LV 5900 (400× to 5000×), to obtain the micrographs.
For the determination of the morphometric parameters of the resting stages, they were photographed in an optical microscope (Nikon Model E600) with a camera (Moticam 2000 2.0M) and later, the images were measured using specialized software (Motic Images plus 2.0). The Maximum Length (ML) and Width (W) were measured following Sarma and Rao (1987). Using the morphometric values, the biovolume of the DE's were calculated through the equations 4/3πr3 for spherical shapes and 4/3πr1r22 for elliptical forms, where r1 is the maximum length and r2 is the width (Walsh et al., 2017).
2.6 Hatching experiments
We conducted two different approximations to estimate the effect of three biotic factors: microalgae, cyanobacteria and kairomones on DE's hatching rate. The first experimental approximation was using specific isolated DE's and the second experiment was conducted with the entire sediment. Both experiments were performed separately for each study site. For each experiment, we set up a total of 27 experimental units (3 sites × 3 treatments × 3 replicates). In addition, we carried out a control group (CTRL) for each site with 3 replicates in which only distilled water was used.
2.7 Biotic factors
All conditioned media were prepared 24 h before being used, and after this time they were filtered through a 0.45 µm Millipore® continuous flow filter. For the conditioned medium of the kairomones treatment (ASP), we used the predatory rotifer Asplanchna girodi which can consume a great variety of rotifers (Conde-Porcuna and Sarma, 1995; Chang, 2010), and has a wide distribution in Mexico (Jiménez-Contreras et al., 2017) including the Tilostoc wetland, from where the strain used in this study was isolated. Individuals of A. girodi were placed in distilled water at a density of 0.1 ind · mL−1 (Aránguiz-Acuña et al., 2010) which is the minimum density necessary for the kairomones of this predator affect their prey and during 24 h because the presence of kairomones can be assured in this time interval (Brönmark and Hansson 2000; Guo et al., 2011). The microalgae conditioned media was prepared by placing the Scenedesmus acutus (SC) microalgae from the University of Texas, at a density of 1 × 106 cell · mL−1 with distilled water, density at which active rotifers develop optimally (Morales-Ventura et al., 2012). Finally, for the conditioned media of cyanobacteria, Microcystis sp. (MC) was isolated from a bloom in Xochimilco Lake, and placed in distilled water at a density of 1 × 106 cell · mL−1, density at which important demographic effects, such as survival and reproduction, on cladocerans and rotifers have been detected (Nandini, 2000).
2.8 Hatching isolated DE's
We selected DE's that were dominant in the three main groups of zooplankton (rotifers, cladocerans and copepods). The same species were placed in equal number and proportion in each experimental unit. The isolation of the resting stages was carried out following the same methodology described for density determination and considering the criteria of healthy DE's described above. Therefore, 10 DE's of rotifers (5 Polyarthra dolichoptera and 5 Keratella tropica), 5 resting stages of cladocerans (Bosmina sp.) and 5 of copepods (Leptodiaptomus sp.) for each experimental unit were placed in culture plates with 1.5 mL of conditioned medium and incubated at 25 °C with constant light (Albritton and White, 2006). The counting of the hatched eggs and the replacement of the corresponding media was done daily for 6 weeks, likewise the newborns were counted and separated. With these data, the hatching rate was calculated.
2.9 Hatching entire sediment
We placed 18 cm−3 of sediment (20 g wet weight), in plastic containers. In order to avoid a possible error associated with the counting of interstitial organisms such as hatching, the sediment was dried in plastic containers at room temperature and covered with a mesh of 20 μm for 2 weeks. After this time, we rehydrated with 100 mL of the corresponding conditioned media for each treatment and incubated at 25 °C with constant illumination to propitiate their hatching. We changed the media daily and monitored the hatching weekly (Albritton and White, 2006) for 6 weeks. The hatched individuals were removed from the experiment and observed in an optical microscope for identification using specialized keys as described above. To prevent the possible permanence of any individual that could reproduce parthenogenically and be counted as a new hatching, the results of this experiment were expressed only as species richness (hatched species).
2.10 Statistical analysis
Normality and homoscedasticity of the hatching rate and number of hatched species (species richness) of the resting stages were assessed with the Shapiro-Wilk and Brown-Forsythe test respectively while statistically significant differences were determined via analysis of variance (one-way ANOVA), followed by a multiple comparison procedure using the Holm-Sidak method. If the normality test failed, a Kruskal-Wallis one-way ANOVA on ranks was performed, all of this in SigmaPlot 11.0 software. EstimateS ver. 9.1.0 was used to obtain Chao 2, and Jackknife 1 species richness estimation rates (Gotelli and Colwell, 2001). These rates were used to observe a possible subestimation of total species richness, considering the incubation time (days) as a sampling effort (Rosa et al., 2022), only in the second hatching experiment (direct hatching from sediments).
3 Results
3.1 Specific richness and density
A total of 22 species of zooplankton were identified. Rotifers had the highest specific richness with 16 species, belonging to 15 genera and 10 families. Cladocerans were represented by 4 species: Macrothrix sp., Bosmina sp., Daphnia parvula and Ceriodaphnia sp. Finally, only one species of copepod, Leptodiaptomus sp. was observed (Tab. 1). The zooplankton egg bank had an average density of 7.6 ± 2 resting stages · cm−3 (Fig. 2). The highest density was observed in PZ (12.4 ± 1.2 resting stages · cm−3) and the lowest in LZ (4.8 ± 1.5 resting stages · cm−3). Rotifers were the zooplankton group with the highest DE's abundance in the three sites (Fig. 2), with a general average of 4.5 ± 1.5 diapausing eggs · cm−3 (P ≤ 0.001, one-way ANOVA, Holm-Sidak, Tab. 2).
In the rotifer DE's, micrographs obtained by light microscopy, it was possible to observe three characteristics, color, shape, and presence of extraembryonic space. The color of all resting stages was brown, and a variable number of globular formations with amber coloration were observed embedded in the embryonic mass. These globular formations were evident in Morphotypes 2, 3 and in Polyarthra dolichoptera (Figs. 3.2C, 3.3C and 3.4C). The predominant shape in the DE's was oval, except for Morphotype 1 that presented a rounded shape (Fig. 3.1). The extraembryonic space was evident only in two DE's, in Morphotype 1 and Ptygura pilula, in the first case was small (1/8 of the whole embryo), while in the second was large (1/2 of the whole embryo) (Figs. 3.1 and 3.3). On the other hand, except in the case of P. pilula eggs, where longitudinal wrinkles were evident, light microscopy did not show the details of the resting stages ornamentation.
The SEM analysis allowed us to the ultrastructure of the seven dominant rotifer DE's. The diapausing egg (DE) identified as Morphotype 1 was the only one that didn't present any external ornamentation (Fig. 3.1A, C), while the other observed DE's were highlighted by a cover sculpted with various ornamentations. Spines were observed in DE's Morphotype 2 and P. dolichoptera. Processes were between 2 and 2.6 µm in height respectively, which were distinguished because in the case of Morphotype 2 they were spike-shaped, whereas in P. dolichoptera were structures with a cylindrical appearance (Figure 3.2A, C and 3.4A, B). Wrinkles were observed in three DE's: Morphotype 3, P. pilula, and T. similis which were identified as pronounced geometrical (hexagonal), concentric and smooth reticular forms respectively (Fig. 3.3A, B, 3.5A, B, C and 3.6A–C). Finally, the outer layer of the Sinantherina DE's was characterized by the presence of holes along the entire surface only interrupted by a transverse suture 0.6 µm wide (Fig. 3.7A, B).
The width of rotifers DEs varied between 47.7 and 84.9 µm, while maximum length between 74 and 195.8 µm. Bosmina resting stages >400 µm wide, but the length of the embryos were 222 µm. The average volume of rotifer DEs was 20 × 105 µm3, T. similis with the smallest volume 7.06 ± 0.001 × 105 µm3 and A. girodi with the highest volume 39.4 ± 0.006 × 105 µm3. The volume of the Bosmina embryos and the resting stages of Leptodiaptomus was 153.7 and 56.1 × 105 μm3 respectively (Tab. 3).
Zooplankton species richness from sediments of Tilostoc wetland.
 |
Fig. 2 Diapause egg density of the three main zooplankton groups (rotifers, cladocerans, and copepods) at the three study sites. |
Results of one-way analysis of variance (ANOVA) performed for the diapausing egg density.
 |
Fig. 3 Rotifer diapausing eggs micrographs with optical and electron microscope (SEM). Numbers within figure show species (1) Morphotype 1, (2) Morphotype 2, (3) Morphotype 3, (4) Polyarthra dolichoptera, (5) Ptygura pilula, (6) Trichocerca similis and (7) Sinantherina sp., and letters show A = Global structure view of diapausing egg whit SEM analysis, B = Zoom on interesting feature/alternative global view SEM analysis, and C = Light microscopy view. |
Morphometry data of dominant resting stages in sediment samples. Volume (1 × 105 μm3 ± SE), ML = maximum length, and W = width (μm ± SE).
3.2 Hatching experiments
According to the first approach used in the experiments with isolated resting stages, the cumulative hatching curve was saturated after 15 days of incubation, after which no hatching was recorded. In these experiments the DE's of rotifers did not hatch, but they were considered in calculating of the hatching rates of Bosmina and Leptodiaptomus. The range of hatching percentage was between 8.3% in the control groups and 30% in the MC treatments yet. The MC treatment showed the highest hatching percentage (30%) and the only one that exceeded the hatching average (21%) of the treatments at the three sites (Tab. 4), however, only on MZ there were significant differences between the treatments (P = 0.047, one-way ANOVA, Kruskal-Wallis, Tab. 5). Particularly, the hatching rate of the resistance structures of the Leptodiaptomus copepod was higher in all treatments except in the control group, where Bosmina had the highest percentages (Tab. 6).
According to the second approach used in the experiments, with entire sediment, 22 species hatched. Some species hatched only in one treatment, for instance, Keratella tropica, Monommata sp., Ptygura pilula and Sinantherina sp. which only appeared in MC, Lindia sp. and Collotheca sp. in the ASP treatment and finally, Anuraeopsis fissa in the CTRL treatment (Tab. 7). On the other hand, some species hatched in all treatments, such as Lecane bulla and Lecane inermis (Tab. 7). The treatment with the highest species richness was MC with 11.3 ± 2, while the lowest value was CTRL with 6.3 ± 0.7 (Fig. 4), nevertheless, no significant differences were found between the treatments. The maximum values of species richness in the different treatments were reached in the sixth week in the three study sites, however, PZ showed the lowest values 4 ± 1.1 of species richness (Appendix A). The models used (Chao 2 and Jackknife 1) for the calculation of theoretical richness in this experiment, showed the same values of observed (Sobs) and theoretical richness in most of the treatments. The only difference was found in the ASP and SC treatments, where the theoretical richness value (8.83 and 7.83) was higher than the observed richness (8 and 7) (Fig. 5).
Hatching rate ± SE (%) of zooplankton resting stages isolated in different sample sites, exposed to three conditioned medium: ASP = Asplanchna, SC = Scenedesmus and MC = Microcystis.
Results of one-way analysis of variance (ANOVA) performed for the hatching rate of zooplankton resting stages isolated and exposed to three conditioned medium.
Hatching rate ± SE (%) of Leptodiaptomus sp. and Bosmina sp. resting stages isolated in different sample sites, exposed to three conditioned medium: ASP = Asplanchna, SC = Scenedesmus and MC = Microcystis.
Occurrence of zooplankton species hatched from sediments, exposed to different conditioned medium (ASP = Asplanchna, SC = Scenedesmus and MC = Microcystis).
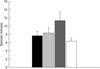 |
Fig. 4 Total rotifer species richness hatched from sediments of Tilostoc wetland, exposed to different treatments (ASP = Asplanchna, black, SC = Scenedesmus, light grey, MC = Microcystis, dark grey, and CTRL = Control, white). |
 |
Fig. 5 Rotifer species accumulation curves from incubated sediments, exposed to different treatments (ASP = Asplanchna, SC = Scenedesmus and MC = Microcystis). Sobs = observed species richness and predicted species richness (Chao2 and Jacknife). |
 |
Fig. A1 Rotifer accumulated species richness ± SE hatched from sediments of Tilostoc wetland, exposed to different treatments (ASP = Asplanchna, SC = Scenedesmus and MC = Microcystis). The dotted line represents the media of data. |
4 Discussion
Taxonomic analyses after sediment hatching was used by May (1986), and has been used since then in some studies (Albritton and White, 2004; Vandekerkhove et al., 2005a, García-Roger et al., 2008) without being adopted as an indispensable tool for diversity work. However, it constitutes an alternative and/or complementary tool in the general detection of the structural patterns of the community in a water body (Gerhard et al., 2016). Our results support the idea of the incorporation of these as a routine analysis in zooplankton diversity studies. The 22 species found by the hatching of the DE's is close to the one obtained in other widely studied adjacent water bodies, such as the Valle de Bravo reservoir, where 23–27 species of rotifers and 7 species of cladocerans have been reported (Nandini et al., 2008; Jiménez-Contreras et al., 2009). It should be noted that the aforementioned studies were carried out over a year and at different depths, whereas the present study represents the species richness from just one collection. Nonetheless, its potential use as a tool for studying diversity will also be limited by the density of DE's in the sediments.
The density of DE's in lacustrine sediments does not show a very definite trend since the range of variation reported ranges from a few to 300 diapausing eggs · cm−3 (Walsh et al., 2017), or a few thousand to millions in a square meter (Hairston, 1996). The density recorded in the Tilostoc wetland was within that interval. However, densities of DE's in Tilostoc were close to the lower limit in the three study sites. This trend can be explained by the fact that we only recorded the densities of the healthy-looking DE's. In Spanish ponds García-Roger et al. (2006) investigated the density of DE's and the fraction corresponding to healthy-looking ones presented values lower than 4 eggs · cm−3 in most systems. Although in deep and eutrophic systems Duggan et al. (2002) obtained densities of up to 300 diapausing eggs · cm3, in several studies carried out on other zooplankton a lower density of DE's has been observed in shallow bodies of water (Carvalho et al., 1989). These wide variations in the density of the DE's in the sediment are mainly related to three factors: the number of DE's that were produced, the number of DE's that had hatched, and their mortality in the sediment (Cáceres and Hairston, 1998; Brendonck and De Meester, 2003). Therefore, an aquatic system where hatching is favored (e.g. a shallow lake, with high light penetration) may have a low density of resting stages, which does not mean that they may not occur.
The different zones of this shallow lake showed slight variations concerning species richness and resting stages densities in the sediment egg bank, where the rotifers are the dominant group. The interaction of intrinsic and extrinsic factors in the lake can explain this trend. The rotifer DE's dominance in the resistance egg bank has been frequently observed in water bodies worldwide with different limnological characteristics (García-Roger et al., 2006; Ning and Nielsen, 2011; Santangelo et al., 2011a; Piscia et al., 2012; Araújo et al., 2013). For example, in Brazilian aquatic systems, Santangelo et al. (2015) obtained a rotifer DE's richness three times greater than cladocerans (62 and 23 species, respectively). Three main reasons might be responsible for explaining the dominance of rotifers. First, rotifers have a higher DE production rate (Gyllström and Hansson, 2004). The second reason is that rotifers can use biotic and abiotic signals to induce diapause over cladocerans and copepods, which require more specific signals (Reviewed in Fryer, 1996; Reviewed in Gyllström and Hansson, 2004). Finally, the resting egg banks reflect the active zooplankton community in the water column (Duggan et al., 2002; Vandekerkhove et al., 2005c).
The morphology of resting stages has been suggested as a reliable, economic and an accessible strategy for the study of the resistance egg bank (Duggan et al., 2002; Vandekerkhove et al., 2004). Although molecular tools have been considered the best approaches in the study of the egg bank (Moreno et al., 2017), they are still expensive, and require the development of sufficiently robust databases to make comparisons, so the morphology can continue to be a good alternative (Segers, 2008; Guerrero-Jiménez et al., 2019). However, the still confusing descriptions of the ornamentation of the resting stages must be overcome (Walsh et al., 2017). One of the most recent and complete works on the morphology of resting stages (Guerrero-Jiménez et al., 2019) highlights the importance of improving the descriptions of resting stages using light microscopy and scanning electron microscopy. For these authors, characteristics such as color and shape were not as informative as the ornamentation of the outer layer of the resting eggs, in determining the species, which is why they highlight the species-specific character of the outer layer. Our data confirm the importance of this external layer in determining the species, as well as the need to combine light microscopy and scanning electron microscopy in their study. By combining these tools, this work provides the descriptions and characteristics of 7 resting stages of rotifers, not included in Guerrero-Jiménez et al. (2019).
The resting stages that we reported as Morphotypes (MT 1–3) did not hatch, so they were identified based on their morphological characteristics. The outer layer of the Morphotype 1 has a smooth surface so it possibly belongs to the genera Synchaeta (Gilbert and Schreiber, 1998, Sarma, pers. Com.). Additionally, in this genera the production of asexual diapause eggs which do not hatch (Gilbert and Schreiber, 1998) has been observed; this may be the reason for its low hatching success. The presence of processes in the form of thorns in the outer layer of the resting stages identified as Morphotype 2, as well as the size of their resting stages, conform to the descriptions made for the Keratella genera. These characteristics and their emergence in the experiments where the complete sediment was used, leads us to the conclusion that they belong to Keratella. Finally, Morphotype 3 is referred to Polyarthra vulgaris (Segers pers. Com.) due to the cylindrical structures in its outer layer that corresponded with the specialized keys (Koste, 1978) and with other images and descriptions (Van Geel, 2001).
Our values in the percentage of hatching (healthy-looking DE's) were equal or less than 30% in all treatments, so they can be considered low if we compare them with those obtained in works directed at aquaculture production (Hagiwara, 1997). However, in most of the works carried out with sediments of natural systems, the resting stages have low hatching rates that range between 6% and 30% (Mugrabe et al., 2007; Santangelo et al., 2011b), which corresponds to our results. These low percentages can be attributed to the genotypic variations that regulate the diapause time, so it can be understood as diverse evolutionary strategy (Hairston et al., 1995). The fact that the MC and SC treatments obtained greater species richness and hatching rate may be related to the ability of phytoplankton to eliminate CO2 and produce oxygen increasing its concentration in water (Jin et al., 2006). Higher of dissolved oxygen concentrations are known to increase the viability time and the hatching rate of the resting stages of zooplankton (Pourriot and Snell, 1983; Lutz et al., 1992). In other studies, where the diapausing eggs of B. plicatilis were exposed to Chlorella stigmatophora, the hatching range has been increased, also attributing it to the variations generated by chlorophyll and dissolved gases (Minkoff et al., 1983). In the same way Broman et al. (2015) obtained a growth in the hatching rates of resting stages from sediments in treatments where oxygenation was increased.
Because each species and population have different signals to break dormancy and end diapause (Brendonck and De Meester, 2003; García-Roger et al., 2008; Radzikowski et al., 2018), generating different responses at hatching, we expected to find significant differences in species richness between treatments. However, this expectation was not confirmed. It has been documented that the similarity in the species richness of rotifers and cladocerans in the egg bank can be high even between lakes in the same region (Santangelo et al., 2015). Recently Yin et al. (2021) observed that biotic factors such as Asplanchna kairomones could be detected by DE's producing early and synchronous hatching patterns. However, when exposure to biotic factors does not explain the variations, these can be understood by the maternal environmental effect during the formation of DE's. The DE's used in this study were obtained from the field, so the maternal environmental signal during its formation is unknown. However, it could have influenced the hatching patterns observed.
On the other hand, it has been suggested that in systems with narrow variations in environmental variables that typically function as a signal for hatching of resting stages (e.g. photoperiod, temperature), zooplankton employ biotic factors (infochemicals, kairomonas), alone or in combination with environmental factors, as signs to break dormancy (reviewed in Fryer, 1996; Brendonck and De Meester, 2003; Reviewed in Gyllström and Hansson, 2004). This idea is also supported by the fact that rotifers and cladocerans use biotic factors as signs of sleep onset (Cáceres and Schwalbach, 2001; Reviewed in Gyllström and Hansson, 2004). However, the greater ease of testing environmental factors has decreased attention to biotic factors such as signs of ending of dormancy (Reviewed in Gyllström and Hansson, 2004). But this lack of attention does not mean that biotic factors do not have an effect, as shown in our results. Likewise, the still incipient and to some extent contradictory results (Lass et al., 2005; Bozelli et al., 2008; Santangelo et al., 2010) on the effect of biological factors on hatching, make it necessary to direct more efforts on this topic.
The hatching rates obtained in this study was low, with a maximum value of 30%, being the MC and SC, the treatments better represented attributed to their capacity of carbon fixation and water oxygenation. We suggest that some species possibly require more dissolved oxygen level to hatch, in comparison to others. The micrographs obtained in this study give a more comprehensive panorama of the different ornamentations of resting stages, contribute to the picture bank, and will allow the taxonomic identification of species based on egg morphology.
Acknowledgments
We thank anonymous reviewers for constructive comments on our manuscript. JJC received SNI support (CVU: 221136) from CONACyT and GZJ has received scholarship (17BBTL0123) from COMECyT (Mexico). We thank Rafael Quintanar-Zuñiga for SEM analysis. We thank S.S.S. Sarma and Nandini S. for suggestions and constructive comments on our manuscript.
Appendix
 |
Fig. A1 Rotifer accumulated species richness±SE hatched from sediments of Tilostoc wetland, exposed to different treatments (ASP=Asplanchna, SC=Scenedesmus and MC=Microcystis). The dotted line represents the media of data. |
References
- Albritton CJ, White DS. 2004. Hatching of rotifer eggs from reservoir sediment. Southeastern Nat 3: 359–370. [CrossRef] [Google Scholar]
- Albritton CJ, White DS. 2006. Rotifer hatching from the sediments of a fluctuating mainstem reservoir. Southeastern Nat 5: 345–354. [CrossRef] [Google Scholar]
- Alekseev VR, Ravera O, De Stasio BT. 2007. Introduction to diapause. In: Alekseev VR, De Stasio BT, Gilbert JJ (eds.), Diapause in aquatic invertebrates: Theory and human use. Dordrecht: Springer, pp. 3–10. [CrossRef] [Google Scholar]
- Aránguiz-Acuña A, Ramos-Jiliberto R, Nandini S, Sarma SSS, Bustamante RO, Toledo V. 2010. Benefits, costs and reactivity of inducible defences: an experimental test with rotifers. Freshw Biol 55: 2114–2122. [CrossRef] [Google Scholar]
- Araújo LR, Lopes PM, Santangelo JM, Petry AC, Bozelli RL. 2013. Zooplankton egg banks in permanent and temporary tropical aquatic systems. Acta Limnol Bras 25: 235–245. [CrossRef] [Google Scholar]
- Balompapueng MD, Munuswamy N, Hagiwara A, Hirayama K. 1997. Effect of disinfectants on the hatching of marine rotifer resting eggs Brachionus plicatillis Müller. Aquac Res 28: 559–565. [CrossRef] [Google Scholar]
- Bhusnale R, Marathe A, Kulkarni MR, Padhye SM, Pai K. 2016. Observations on the hatching dynamics and phenology of co-occurring large branchiopods from a small temporary pool in western India using ex situ sediment rehydration. Ann Limnol Int J Limnol 52: 327–333. [CrossRef] [EDP Sciences] [Google Scholar]
- Bozelli RL, Tonsi M, Sandrini F, Manca M. 2008. A big bang or small bangs? Effect of biotic environment on hatching. J Limnol 67: 100–106. [CrossRef] [Google Scholar]
- Brandão LPM, Pujoni DGF, Maia-Barbosa PM. 2014. Seasonal dynamics of Daphnia laevis Birge, 1878. Ephippia in a tropical lake with a description of a new methodology for in situ evaluation. Braz J Biol 43: 642–648. [CrossRef] [PubMed] [Google Scholar]
- Brendock L, De Meester L. 2003. Egg bank of the freshwater zooplankton: evolutionary and ecological archives in the sediment. Hydrobiologia 491: 65–84. [CrossRef] [Google Scholar]
- Brock MA, Nielsen DL, Crosslé K. 2005. Changes in biotic communities developing from freshwater wetland sediments under experimental salinity and water regimes. Freshw Biol 50: 1376–1390. [Google Scholar]
- Broman E, Brüsin M, Dopson M, Hylander S. 2015. Oxygenation of anoxic sediments triggers hatching of zooplankton eggs. P R Soc B 282: 2015–2025. [Google Scholar]
- Brönmark C, Hansson LA. 2000. Chemical communication in aquatic systems: an introduction. Oikos 88: 103–109. [CrossRef] [Google Scholar]
- Cáceres CE, Hairston NG Jr. 1998. Benthic-pelagic coupling in planktonic crustaceans: the role of the benthos. Arch Hydrobiol 52: 163–174. [Google Scholar]
- Cáceres CE, Schwalbach MS. 2001. How well do laboratory experiments explain field patterns of zooplankton emergence? Freshw Biol 46: 1179–1189. [CrossRef] [Google Scholar]
- Conde-Porcuna JM, Perez-Martinez C, Moreno E. 2018. Variations in the hatching response of rotifers to salinity and waterbird ingestion. J Plankton Res 40: 326–341. [CrossRef] [Google Scholar]
- Conde-Porcuna JM, Ramos-Rodríguez E, Pérez-Martínez C. 2014. In situ production of empty ephippia and resting eggs by an obligate parthenogenetic Daphnia population. J Plankton Res 36: 157–169. [CrossRef] [Google Scholar]
- Conde-Porcuna JM, Sarma SSS. 1995. Prey selection by Asplanchna girodi (Rotifera): The importance of prey defence mechanisms. Freshw Biol 33: 341–348. [CrossRef] [Google Scholar]
- De Smet WH. 1998. Preparation of rotifer trophi for light and scanning electron microscopy. Hydrobiologia 388: 117–121. [CrossRef] [Google Scholar]
- De Stasio BT. 2007. Egg bank formation by aquatic invertebrates: A bridge across disciplinary boundaries. In: Alekseev VR, De Stasio BT, Gilbert JJ (eds.), Diapause in aquatic invertebrates: Theory and human use. Dordrecht: Springer, pp. 121–133. [CrossRef] [Google Scholar]
- Decaestecker E, De Meester L, Mergeay J. 2009. Cyclical parthenogenesis in Daphnia: sexual versus asexual reproduction. In: Schön I, Martens K and Dijk PV (eds.), Lost sex: The evolutionary biology of parthenogenesis. Dordrecht: Springer, pp. 295–316. [CrossRef] [Google Scholar]
- Dodson SL, Cáceres CE, Rogers DC. 2009. Cladocera and other Branchiopoda. In: Thorp JH, Covich AP (eds.), Ecology and Classification of North American Freshwater Invertebrates. Amsterdam: Elsevier, pp. 773–827 [Google Scholar]
- Duggan IC, Green JD, Shiel RJ. 2002. Rotifer resting egg densities in lakes of different trophic state, and their assessment using emergence and egg counts. Arch Hydrobiol 153: 409–420. [CrossRef] [Google Scholar]
- Elías-Gutiérrez M, Suárez-Morales E, Gutiérrez-Aguirre MA, Silva-Briano M, Granados-Ramírez JG, Garfias-Espejo T. 2008. Cladocera y copepoda de las aguas continentals de México: Guía ilustrada. UNAM, CONABIO, ECOSUR, CONACyT, SEMARNAT, México DF-México. [Google Scholar]
- Fontaneto D, De Smet WH. 2015. Rotifera. In: Schmidt-Rhaesa (ed.), Handbook of zoology: Gastroticha, Cycloneuralia and Gnatifera vol 3 Gastrotricha and Gnatifera. Berlin: De Gruyter, pp. 217–300. [Google Scholar]
- Fryer G. 1996. Diapause, a pontent forcé in the evolution of freshwater crustaceans. Hydrobiologia 320: 1–14. [CrossRef] [Google Scholar]
- García-Roger EM, Armengol-Díaz X, Carmona MJ, Serra M. 2008. Assessing rotifer diapausing egg bank diversity and abundance in brackish temporary environments an ex situ sediment incubation approach. Arch Hydrobiol 173: 79–88. [CrossRef] [Google Scholar]
- García-Roger EM, Carmona MJ, Serra M. 2006. Patterns in rotifer diapausing egg banks: density and viability. J Exp Mar Biol Ecol 336: 198–210. [CrossRef] [Google Scholar]
- Gerhard M, Iglesias C, Clemente JM, Goyenola G, Meerhoff M, Pacheco JP, Teixeira-De Mello F, Mazzeo N. 2016. What can resting egg banks tell about cladoceran diversity in a shallow subtropical lake? Hydrobiologia 798: 75. [Google Scholar]
- Gilbert JJ, Schreiber DK. 1998. Asexual diapause induced by food limitation in the rotifer Synchaeta pectinata. Ecology 79: 1371–1381. [CrossRef] [Google Scholar]
- Gilbert JJ, Wurdak ES. 1978. Species-specific morphology of resting eggs in the rotifer Asplanchna. T Am Microsc Soc 97: 330–339. [CrossRef] [Google Scholar]
- Gilbert JJ. 1974. Dormancy in rotifers. T Am Microsc Soc 93: 490–513. [CrossRef] [Google Scholar]
- Gilbert JJ. 1977. Mictic-female production in monogonont rotifers. Arch Hydrobiol Beih Ergebn Limnol 8: 142–155. [Google Scholar]
- Gilbert JJ. 2003. Specifity of crowding response that induces sexuality in the rotifer Brachionus. Limnol Oceanogr 48: 1297–1303. [CrossRef] [Google Scholar]
- Gilbert JJ. 2007. Timing of diapause in monogonont rotifers. Mechanisms and strategies. In: Alekseev VR, De Stasio BT, Gilbert JJ (eds.), Diapause in aquatic invertebrates: Theory and human use. Dordrecht: Springer, pp. 11–27. [CrossRef] [Google Scholar]
- Gilbert JJ. 2010. Effect of food concentration on the production and viability of resting eggs of the rotifer Brachionus: implications for the timing of sexual reproduction. Freshw Biol 55: 2437–2446. [CrossRef] [Google Scholar]
- Gilbert JJ, Schröder T. 2004. Rotifers from diapausing, fertilized eggs: unique features and emergence. Limnol Oceanogr 49, 1341–1354. [CrossRef] [Google Scholar]
- Gill HK, Goyal G, Chahil G. 2017. Insect diapause: a review. J Agric Sci Technol 7: 454–473. [Google Scholar]
- Gotelli NJ, Colwell RK. 2001. Quantifying biodiversity: procedures and pitfalls in the measurement and comparison of species richness. Ecol Lett 4: 379–391. [CrossRef] [Google Scholar]
- Guerrero-Jiménez G, Ramos-Rodríguez E, Silva-Briano M, Albadache-Ortíz A, Conde-Porcuna JM. 2019. Analysis of the morphological structure of diapausing propagules as a potential tool for the identification of rotifer and cladoceran species. Hydrobiologia 847: 243–266. [Google Scholar]
- Guo R, Snell TW, Yang J. 2011. Ecological strategy of rotifer (Brachionus calyciflorus) exposed to predator- and competitor-conditioned media. Hydrobiologia 658: 163–171. [CrossRef] [Google Scholar]
- Gyllström M, Hansson LA. 2004. Dormancy in freshwater zooplankton: induction, termination and the importance of benthic-pelagic coupling. Aquat Sci 66: 274–295. [Google Scholar]
- Hagiwara A, Balompapueng MD, Munuswamy N, Hirayama K. 1997. Mass production and preservation of the resting eggs of the euryhaline rotifer Brachionus plicatilis and B rotundiformis. Aquaculture 155: 223–230. [CrossRef] [Google Scholar]
- Hairston NG. 1996. Zooplankton egg banks as biotic reservoirs in changing environments. Limnol Oceanogr 41: 1087–1092. [CrossRef] [Google Scholar]
- Hairston GN, Fox JA. 2009. Egg banks. In: Gene EL (ed.), Encyclopedia of Inland Waters Amsterdam: Elsevier, pp. 659–666. [CrossRef] [Google Scholar]
- Hairston GN, Van Brunt JRA, Kearns CM. 1995. Age and survivorship of diapausing eggs in a sediment egg bank. Ecology 76: 1706–1711. [CrossRef] [Google Scholar]
- Hairston NG, Hansen AM, Schaffner WR. 2000. The effect of diapause emergence on the seasonal dynamics of a zooplankton assemblage. Freshw Biol 45: 133–145. [CrossRef] [Google Scholar]
- Irigoien X, Harris R, Verheye H, Joly P, Runge J, Starr M, Pond D, Campbell R, Shreeve R, Ward P, Smith AN, Dam HG, Peterson W, Tirelli V, Koski M, Smith T, Harbour D, Davidson R. 2002. Copepod hatching success in marine ecosystems with high diatom concentrations. Nature 419: 387–389. [CrossRef] [PubMed] [Google Scholar]
- Jiménez-Contreras J, Sarma SSS, Piedra-Ibarra E, Nandini S. 2017. Morphometric and molecular (COX1) variations of Asplanchna girodi clones from central Mexico. J Environ Biol 38: 1229–1239. [CrossRef] [Google Scholar]
- Jiménez-Contreras J, Sarma SSS, Merino-Ibarra M, Nandini S. 2009. Seasonal changes in the rotifer (Rotifera) diversity from a tropical high altitude reservoir (Valle de Bravo, Mexico). J Environ Biol 30: 191–195. [PubMed] [Google Scholar]
- Jin HF, Lim BR, Lee K. 2006. Influence of nitrate feeding on carbon dioxide fixation by microalgae. J Environ Sci Health Toxic/Hazard Subst Environ Eng 41: 2813–2824. [Google Scholar]
- Kanagasabapathi V, Munuswamy N. 2011. Preservation, development, and hatching of resting eggs in the freshwater rotifer Brachionus calyciflorus Pallas. Microsc Res Techniq 74: 744–748. [CrossRef] [Google Scholar]
- King CE, Snell TW. 1980. Density-dependent sexual reproduction in natural pupulations of the rotifer Asplanchna girodi. Hydrobiologia 73: 149–152. [CrossRef] [Google Scholar]
- Kleiven OT, Lasson P, Hobaek A. 1992. Sexual reproduction in Daphnia magna requires three stimuli. Oikos 65: 197–206. [CrossRef] [Google Scholar]
- Kogane T, Hagiwara A, Imaizumi K. 1997. Temperature conditions enhancing resting egg production of the euryhaline rotifer Brachionus plicatilis O F Muller (Kamiura strain). Hydrobiologia 358: 167–171. [CrossRef] [Google Scholar]
- Koštál V. 2006. Eco-physiological phases of insect diapause. J Insect Physiol 52: 113–127. [CrossRef] [PubMed] [Google Scholar]
- Koste W. 1978. Rotatoria. Die Rädertierte Mitteleuropas. Ein Bestimmungswerk, begründet von Max Voigt. Überordnung, Monogononta 2. Gebrüder Borntraeger, Berlin, Stuttgart, 673. [Google Scholar]
- Lampert W, Sommer U. 2007. Limnoecology: The Ecology of Lakes and Streams. USA: Oxford University Press. [Google Scholar]
- Lass S, Vos M, Wolinska J, Spaak P. 2005. Hatching with the enemy: Daphnia diapausing eggs hatch in the presence of fish kairomones. Chemoecology 15: 7–12. [CrossRef] [Google Scholar]
- Lutz RV, Marcus NH, Chanton JP. 1992. Effects of low oxygen concentrations on the hatching of eggs of marine calanoid copepods. Mar Biol 114: 241–247. [CrossRef] [Google Scholar]
- Maia-Barbosa PM, Eskinazi-Sant'Anna EM, Valadares CF, Pessoa GCD. 2003. The resting eggs of zooplankton from a tropical, eutrophic reservoir (Pampulha Reservoir, south-east Brazil). Lakes Reserv: Res Manag 8: 269–275. [CrossRef] [Google Scholar]
- Marcus NH. 2005. Calanoid copepods, resting eggs, and aquaculture. In: Cheng-Sheng L, O'Bryen PJ, Marcus NH (eds.), Copepods in Aquaculture. Oxford: Blackwell Publishing, pp. 3–9. [CrossRef] [Google Scholar]
- May L. 1986. Rotifer sampling a complete species list from one visit? Hydrobiologia 134: 117–120. [CrossRef] [Google Scholar]
- Minkoff G, Lubzens E, Kahan D. 1983. Environmental factors affecting hatching of rotifer (Brachionus plicatilis) resting eggs. Hydrobiologia 104: 61–69. [CrossRef] [Google Scholar]
- Miralto A, Barone G, Romano G, Poulet S.A., Ianora A, Russo GL, Buttino I, Mazzarella G, Laabir M, Cabrini M, Giacobbe MG. 1999. The insidious effect of diatoms on copepod reproduction. Nature 402: 173–176. [CrossRef] [Google Scholar]
- Morales-Ventura J, Nandini S, Sarma SSS, Castellanos-Páez ME. 2012. Demography of zooplankton (Anuraeopsis fissa, Brachionus rubens and Moina macrocopa) fed Chlorella vulgaris and Scenedesmus acutus cultured on different media. Rev Biol Trop 60: 955–965. [CrossRef] [PubMed] [Google Scholar]
- Moreno E, Pérez-Martínez C, Conde-Porcuna JM. 2016. Dispersal of zooplankton dormant propagules by wind and rain in two aquatic systems. Limnetica 32: 323–336. [MathSciNet] [Google Scholar]
- Moreno E, Conde-Porcuna JM, Gómez A. 2017. Bacording rotifer biodiversity in Mediterranean ponds using diapausing egg banks. Ecol Evol 7: 4855–4867. [CrossRef] [PubMed] [Google Scholar]
- Mowe MA, Mitrovic SM, Lim RP, Furey A, Yeo DC. 2015. Tropical cyanobacterial blooms: a review of prevalence, problem taxa, toxins and influencing environmental factors. J Limnol 74: 205–224. [Google Scholar]
- Mugrabe G, Barro S, Mazzaro A, Valentin JL. 2007. Hatching rates of resting eggs of ‘Cladocera’ (Crustacea; Branchiopoda) at a tropical bay, Brazil. Braz J Biol 67: 527–530. [CrossRef] [PubMed] [Google Scholar]
- Nandini S. 2000. Responses of rotifers and cladocerans to Mycrocistis aeruginosa (Cyanophyceae): a demographic study. Aquat Ecol 34: 227–242. [Google Scholar]
- Nandini S, Merino-Ibarra M, Sarma SSS. 2008. Seasonal changes in the zooplankton abundances of the reservoir Valle de Bravo (State of Mexico, Mexico). Lake Reserv Manag 24: 321–330. [CrossRef] [Google Scholar]
- Ning NSP, Nielsen DL. 2011. Community structure and composition of microfaunal egg bank assemblages in riverine and floodplain sediments. Hydrobiologia 661: 211–221. [CrossRef] [Google Scholar]
- Onbé T. 1978. Sugar flotation method for sorting the resting eggs of marine cladocerans and copepods from sea-bottom sediment. Bull Jpn Soc Sci Fish 44: 1411. [CrossRef] [Google Scholar]
- Paffenhöfer GA. 2002. An assessment of the effects of diatoms on planktonic copepods. Mar Ecol Prog Ser 227: 305–310. [CrossRef] [Google Scholar]
- Piscia R, Guilizzoni P, Fontaneto D, Vignati D, Appleby PG, Manca M. 2012. Dynamics of rotifer and cladoceran resting stages during copper pollution and recovery in a subalpine lake. Ann Limnol. Int J Lim 48: 151–160. [CrossRef] [EDP Sciences] [Google Scholar]
- Portinho JL, Nielsen DL, Daré L, Henry R, Oliveira RC, Branco CCZ. 2018. Mixture of commercial herbicides based on 2,4-D and glyphosate mixture can suppress the emergence of zooplankton from sediments. Chemosphere 203: 151–159. [CrossRef] [PubMed] [Google Scholar]
- Pourriot R, Clément P. 1977. Comparison of the control of mixis in three clones of Notommata copeus. Arch Hydrobiol Beih Ergebn Limnol 8: 174–177. [Google Scholar]
- Pourriot R, Snell TW. 1983. Resting eggs in rotifers. Hydrobiologia 104: 213–224. [CrossRef] [Google Scholar]
- Radzikowski J, Krupińska K, Ślusarczyk M. 2018. Different thermal stimuli initiate hatching of Daphnia diapausing eggs originating from lakes and temporary waters. Limnology 19: 81–88. [CrossRef] [Google Scholar]
- Ramírez-Zierold J, Merino-Ibarra M, Monroy-Ríos E, Olson M, Castillo FS, Gallegos ME, Vilaclara G. 2010. Changing water, phosphorus and nitrogen budgets for Valle de Bravo reservoir, water supply for Mexico City Metropolitan Area. Lake Reserv Manag 26: 23–34. [CrossRef] [Google Scholar]
- Ricci C. 2001. Dormancy patterns in rotifers. Hydrobiologia 446/447: 1–11. [CrossRef] [Google Scholar]
- Rosa J, Martens K, Higuti J. 2022. Dried aquatic macrophytes are floating egg banks and potential dispersal vectors of ostracods (Crustacea) from pleuston communities. Hydrobiologia https://doi.org/10.1007/s10750-022-04818-8 [Google Scholar]
- Santangelo JM, Bozelli RL, Esteves FDA, Tollirian R. 2010. Predation cues do not affect the induction and termination of diapause in small-bodied cladocerans. Freshw Biol 55: 1577–1586. [CrossRef] [Google Scholar]
- Santangelo JM, Esteves FA, Manca M, Bozelli RL. 2011a. Abundance, composition and spatial variation in the egg bank of a tropical zooplankton community. Stud Neotrop Fauna Environ 46: 225–232. [CrossRef] [Google Scholar]
- Santangelo JM, Araujo LR, Esteves FA, Manca M, Bozelli RL. 2011b. Method for hatching resting eggs from tropical zooplankton: effects of drying or exposing to low temperatures before incubation. Acta Limnol Bras 23: 42–47. [CrossRef] [Google Scholar]
- Santagelo JM, Lopes PM, Nascimento MO, Fernandes APC, Bartole S, Figueiredo-Barros MP, Leal JJL, Esteves FA, Farjalla V.F., Bonecker CC, Bozelli RL. 2015. Community structure of resting eggs banks and concordance patterns between dormant and active zooplankters in tropical lakes. Hydrobiologia 758: 183–195. [CrossRef] [Google Scholar]
- Sarma SSS, RAO TR. 1987. Effect of food level on body size and egg size in a growing population of the rotifer Brachionus patulus Müller. Arch Hydrobiol 111: 245–253. [CrossRef] [Google Scholar]
- Segers H. 2008. Global diversity of rotifers (Phylum Rotifera) in freshwater. Hydrobiologia 595: 49–59. [CrossRef] [Google Scholar]
- Shield RJ. 1995. A guide to identification of rotifers, cladocerans and copepods from Australian inland waters. Albury, NSW: Co-operative Research Centre for Freshwater Ecology. [Google Scholar]
- Ślusarczyk M, Flis S. 2019. Light quantity, not photoperiod terminates diapause in the crustacean Daphnia. Limnol Oceanogr 64: 124–130. [CrossRef] [Google Scholar]
- Snell TW, Burke BE, Messur SD. 1983. Size and distribution of resting eggs in a natural population of the rotifer Brachionus plicatilis. Gulf Res Rep 7: 285–287. [Google Scholar]
- Stelzer CP, Lehtonen J. 2016. Diapause and maintenance of facultative sexual reproductive strategies. Philos T Roy Soc B 371: 20150536. [CrossRef] [PubMed] [Google Scholar]
- Taylor F. 1980. Optimal switching to diapause in relation to the onset of winter. Theor Popul Biol 18: 125–133. [CrossRef] [PubMed] [Google Scholar]
- Van Geel B. 2001. Non-pollen palynomorphs. In: Smol JP, Birks HJB, Last WM (eds.), Tracking Environmental Change Using Lake Sediments. Terrestrial, Algal, and Siliceous Indicators. Kluwer Academic Publishers, pp. 99–119. [Google Scholar]
- Vandekerkhove J, Declerck S, Vanhove M, Brendonck L, Jeppensen E, Conde-Porcuna JM, De Meester L. 2004. Use of ephippial morphology to asses richness of anomopods: potentials and pitfalls. J Limnol 63: 75–84. [CrossRef] [Google Scholar]
- Vandekerkhove J, Declerck S, Brendonck L, Conde-Porcuna JM, Jeppesen E, De Meester L. 2005a. Hatching of cladoceran resting eggs: temperature and photoperiod. Freshw Biol 50: 96–104. [CrossRef] [Google Scholar]
- Vandekerkhove J, Declerck S, Brendonck L, Conde-Porcuna JM, Jeppesen E, Johansson LS, De Meester L. 2005b. Uncovering hidden species: hatching diapausing eggs for the analysis of cladoceran species richness. Limnol Oceanogr Meth 3: 399–407. [CrossRef] [Google Scholar]
- Vandekerkhove J, Declerck S, Jeppesen E, Conde-Porcuna JM, Brendonck L, De Meester L. 2005c. Dormant propagule banks integrate spatio-temporal heterogeneity in cladoceran communities. Oecologia 142: 109–116. [CrossRef] [PubMed] [Google Scholar]
- Vargas AL, Santangelo JM, Bozelli RL. 2019. Recovery from drought: viability and hatching patterns of hydrated and desiccated zooplankton resting eggs. Int Rev Hydrobiol 104: 26–33. [CrossRef] [Google Scholar]
- Walsh EJ, Smith HA, Wallace RL. 2014. Rotifers of temporary waters. Int Rev Hydrobiol 99: 3–19. [CrossRef] [Google Scholar]
- Walsh EJ, May L, Wallace RL. 2017. A metadata approach to documenting sex in phylum Rotifera: diapausing embryos, males, and hatchlings from sediments. Hydrobiologia 796: 265–276. [CrossRef] [Google Scholar]
- Yin X, Zhao Y, Tian S, Li X. 2021. Dormant plasticity of rotifer diapausing eggs in response to predator kairomones. Biol Lett 17: 20210422. [CrossRef] [PubMed] [Google Scholar]
- Zeller M, Jiménez-Melero R, Santer B. 2004. Diapause in the calanoid freshwater copepod Eudiaptomus graciloides. J Plankton Res 26: 1379–1388. [CrossRef] [Google Scholar]
Cite this article as: García-Zamorano J, Jiménez-Contreras J. 2023. Zooplankton egg bank: characterization and effect of biotic factors on hatching. Int. J. Lim. 59: 3:
All Tables
Results of one-way analysis of variance (ANOVA) performed for the diapausing egg density.
Morphometry data of dominant resting stages in sediment samples. Volume (1 × 105 μm3 ± SE), ML = maximum length, and W = width (μm ± SE).
Hatching rate ± SE (%) of zooplankton resting stages isolated in different sample sites, exposed to three conditioned medium: ASP = Asplanchna, SC = Scenedesmus and MC = Microcystis.
Results of one-way analysis of variance (ANOVA) performed for the hatching rate of zooplankton resting stages isolated and exposed to three conditioned medium.
Hatching rate ± SE (%) of Leptodiaptomus sp. and Bosmina sp. resting stages isolated in different sample sites, exposed to three conditioned medium: ASP = Asplanchna, SC = Scenedesmus and MC = Microcystis.
Occurrence of zooplankton species hatched from sediments, exposed to different conditioned medium (ASP = Asplanchna, SC = Scenedesmus and MC = Microcystis).
All Figures
 |
Fig. 1 Sample points in Tilostoc wetland (MZ = Macrophyte Zone, PZ = Pelagic Zone, and LZ = Littoral Zone) and its location in Mexico State, Mexico. The grey area represents the part of waterbody with presence of macrophytes of genre Tipha. |
| In the text | |
 |
Fig. 2 Diapause egg density of the three main zooplankton groups (rotifers, cladocerans, and copepods) at the three study sites. |
| In the text | |
 |
Fig. 3 Rotifer diapausing eggs micrographs with optical and electron microscope (SEM). Numbers within figure show species (1) Morphotype 1, (2) Morphotype 2, (3) Morphotype 3, (4) Polyarthra dolichoptera, (5) Ptygura pilula, (6) Trichocerca similis and (7) Sinantherina sp., and letters show A = Global structure view of diapausing egg whit SEM analysis, B = Zoom on interesting feature/alternative global view SEM analysis, and C = Light microscopy view. |
| In the text | |
 |
Fig. 4 Total rotifer species richness hatched from sediments of Tilostoc wetland, exposed to different treatments (ASP = Asplanchna, black, SC = Scenedesmus, light grey, MC = Microcystis, dark grey, and CTRL = Control, white). |
| In the text | |
 |
Fig. 5 Rotifer species accumulation curves from incubated sediments, exposed to different treatments (ASP = Asplanchna, SC = Scenedesmus and MC = Microcystis). Sobs = observed species richness and predicted species richness (Chao2 and Jacknife). |
| In the text | |
 |
Fig. A1 Rotifer accumulated species richness ± SE hatched from sediments of Tilostoc wetland, exposed to different treatments (ASP = Asplanchna, SC = Scenedesmus and MC = Microcystis). The dotted line represents the media of data. |
| In the text | |
 |
Fig. A1 Rotifer accumulated species richness±SE hatched from sediments of Tilostoc wetland, exposed to different treatments (ASP=Asplanchna, SC=Scenedesmus and MC=Microcystis). The dotted line represents the media of data. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


