| Issue |
Int. J. Lim.
Volume 59, 2023
|
|
|---|---|---|
| Article Number | 9 | |
| Number of page(s) | 14 | |
| DOI | https://doi.org/10.1051/limn/2023007 | |
| Published online | 02 August 2023 | |
Research article
Use of different indices to assess the ecological status of lake systems in the eastern mediterranean river basin
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
22
November
2022
Accepted:
21
June
2023
Abstract
The objective of this research was to assess the ecological status of lentic systems in the Eastern Mediterranean River basin (Türkiye) using some biotic (Q index, PT-BV, MedPTI, TDIL, and PTI) and abiotic (WQI, WQImin-nw, TSI, TLI, and Kna) indices. Phytoplankton species such as Peridinium cinctum, Ceratium hirundinella, and Gyrosigma balticum were the species that contributed the most to the algal biovolume of lake systems with different ecological statuses in the basin. According to the results, it was seen that CCA coordination, which analyzed the relationship structures between dominant phytoplankton taxa and environmental water quality parameters, explained the variation sufficiently. As pointed out in the CCA analysis, conductivity and ammonium were the main environmental parameters influencing algal assemblages at sampling sites in the basin (p < 0.01). Strong correlations were observed between TSI and TLI (correlation coefficient: 0.99), and TDIL showed significant correlation only with the Q index (p < 0.01). TSI, TLI and WQI, abiotic indices, indicated significant correlations with most environmental parameters (p < 0.01), while PTI, a biotic index, had weak correlations with most environmental parameters (p > 0.05). Among the indices used in this study, it seems that diatom-based TDIL and physicochemical-based WQI appear to be the most suitable indices for assessing the ecological status of lentic systems in the Mediterranean region. Accordingly, it can be deduced that coupling biotic and abiotic indices is more accurate in determining the water quality of lentic systems.
Key words: Biotic assessment / Abiotic assesment / Phytoplankton / Diatoms / River Basin
© EDP Sciences, 2023
1 Introduction
Phytoplankton are tiny floating algae that are sensitive to nutrient levels and can cause problems like algal blooms, and an imbalance in water ecosystems if there are too many nutrients. Excess nutrients can create eutrophication, a process characterised by increased plant growth, problematic algal blooms and undesirable disturbance of the balance of organisms present in the water (EEA, 2018). The Water Framework Directive (WFD) (EC, 2000) requires achieving good status in all water bodies, and phytoplankton is an important biological quality element that must be monitored for the ecological status assessment in lakes and reservoirs according to the WFD. Diatoms are a major component of the phytobenthos in lotic and lentic systems (Bennion et al., 2014) and should also be monitored in WFD assessments (EC, 2000).
The phytoplankton quality element should be assessed in terms of composition, biovolume, and abundance for the ecological status assessment. Composition indices especially use total phosphorus as a proxy in PTI (Phytoplankton Trophic Index) (Phillips et al., 2013) and MedPTI (Mediterranean Phytoplankton Trophic Index) (Marchetto et al., 2009). The PTI index derived from 20 European countries is widely used in European lakes (Molina-Navarro et al., 2014; Carvalho et al., 2013; Lyche-Solheim et al., 2013). The MedPTI index is also a useful tool to verify the impact of eutrophication in Mediterranean basin. Many researches in Europe have employed the MedPTI index (Vadrucci et al., 2017; Molina-Navarro et al., 2014). The assemblage index (Q index) based on phytoplankton functional groups, developed by Padisák et al. (2006) was used to assess the ecological status of lakes. The Q index has a strong theoretical foundation and can be utilized to assess ecological status across different regions. It has been extensively employed in studies of European lakes, as demonstrated by Pasztaleniec and Poniewozik (2010) and Poniewozik and Lenard (2022). In recent years, a large number of monitoring activities have been carried out in Türkiye, and phytoplankton biovolume (PT-BV) with cyanobacteria biovolume (CY-BV) indices were developed (DGWM, 2020). The ecological status of deep and shallow lakes was classified according to total and cyanobacteria biovolume. Many European phytoplankton indices have been studied in Turkish lakes (Demir et al., 2014; Sevindik et al., 2017; Çelekli et al., 2018, Çelekli et al., 2020). The Phytobenthos quality element should also be evaluated in the littoral regions of lakes. The Trophic Diatom Index for Lakes (Stenger-Kovacs et al., 2007) was developed as a trophic diatom index on the basis of diatom species' optimum and tolerance charecteristics along the total phosphorus gradient in Hungary.
In addition to using biological indices, researchers also investigated water quality using environmental factors. Some trophic indices, such as Carlson (1977), Walker (1982), and Burns et al. (1999), use eutrophication-related parameters like total phosphorus, total nitrogen, chlorophyll a, and transparency. Other water quality indices, such as Ramakrishnaiah et al. (2009) and Wu et al. (2021), incorporate additional parameters such as pH, electrical conductivity, dissolved oxygen, total dissolved solids, calcium, magnesium, chloride, sulfate, nitrate, ammonium, biological oxygen demand, chemical oxygen demand, total hardness, bicarbonate, iron, manganese, fluorides, sulphur, mercury, zinc, and cadmium. The Carlson trophic index is frequently utilized to assess lakes in Europe and Turkey (Jarosiewicz et al., 2011; Kutlu et al., 2017; Jekatierynczuk-Rudczyk et al., 2014). Water quality evaluations with environmental parameters can be done more quickly and simply according to biotic indices.
The Eastern Mediterranean River basin is found in the south of Türkiye and flows its waters into the Mediterranean Sea. While the point sources of pollution in the Eastern Mediterranean basin are urban and industrial wastewater, agriculture and livestock activities are among the diffuse sources (DGWM, 2013). In a study carried out in the basin, Alp et al. (2016) determined the water quality and trophic status of Akgöl as a mesotrophic structure. Koyuncu and Çevik (2014) indicated that the Berdan Dam in the basin belong to the mesotrophic class according to phytoplankton composition. In this study, we aimed (1) to check the correlations between environmental water quality parameters (pH, water temperature, dissolved oxygen, conductivity, ammonium-nitrogen, nitrite, nitrate, sulfate, suspended solids, biochemical oxygen demand, chemical oxygen demand, total organic carbon, total nitrogen, total phosphorus), phytoplankton species, biotic (Q index, PT-BV, MedPTI, TDIL, and PTI) and abiotic (WQI, WQImin-nw, TSI, TLI, and Kna) indices. (2) According to correlations, we tried to evaluate the ecological status of lentic systems in the Eastern Mediterranean River basin (Türkiye).
2 Materials and methods
2.1 Study area
The Eastern Mediterranean River basin is one of 25 basins in Türkiye, and with an area of 2.18 × 104 km2. The basin is located within the geographical co-ordinates 36°00′−37°28′N and 32°06′−35°09′E (DGWM, 2016). The basin area includes the water collection areas of the rivers between the Sedir River in the west and the Tarsus River in the south of Türkiye, and covers the area that discharges the water of the Göksu River (Bayer-Altin and Altin, 2021). Phytoplankton and diatoms were sampled twice (autumn and spring) a year during 2020 and 2021 in each lake. The eighteen monitoring stations covering 7 lakes were carefully selected to represent the Eastern Mediterranean River basin. The lakes considered in the study area were illustrated in Figure 1, and features of the lakes were given in Table 1.
 |
Fig. 1 Location map of the study area in the Eastern Mediterranean River basin. |
Sampling sites of the Eastern Mediterrenian River basin.
2.2 Sampling and field analysis
Water temperature (Temp), pH, dissolved oxygen (DO), and conductivity (EC) were measured in situ with electronic field probes (YSI, model Pro Plus). Water samples were collected and then transported to the laboratory in cool boxes and analyzed according to standard methods (APHA, 2012) within 24 h. The concentrations of ammonium-nitrogen (NH4+-N), nitrite (NO2-N), nitrate (NO3-N), sulfate (SO4), total nitrogen (TN), and total phosphorus (TP) were quantified. Suspended solids (SS), biological oxygen demand (BOD5), chemical oxygen demand (COD), and total organic carbon (TOC) were also determined. All described parameters were obtained seasonally (autumn 2020 and spring 2021) from seven lakes (L1-L7) whose coordinates and monitoring station numbers are given in Table 1.
2.3 Laboratory analysis
The phytoplankton samples were collected with a Hydrobios water sampler, and a plankton net with a pore diameter of 50 μm was also used for collecting samples of phytoplankton. Diatom samples were obtained in the littoral zone of the lakes, near the stations that were selected for phytoplankton sampling. Epiphytic diatoms were preferred, however, if macrophytes such as Typha or Phragmites were absent, epilithic diatoms were sampled. All the samples (phytoplankton and diatoms) were fixed by an acetic Lugol solution (Sournia, 1978). The diatom samples were cleaned with hydrochloric acid and hot hydrogen peroxide (EC, 2014), and permanent slides were mounted with Naphrax.
Phytoplankton were enumerated from a 10 ml sub-sample after settling for 24 h in an Utermöhl style counting chamber. The phytoplankton counts were made in the inverted microscope from the entire chamber following the Utermöhl methodology (Utermöhl, 1958), and the diatom frustules were counted and identified by permanent slides using a light microscope (630× and/or 1000× magnification). The total biovolume of the phytoplankton samples was estimated using biovolume estimation techniques (Hillebrand et al., 1999; Sun and Liu, 2003) and calculated using the corresponding geometrical forms. The taxa identification for phytoplankton and diatom, the most common taxonomic literature was applied (Krammer and Lange Bertalot, 1991a; 1991b; 1999a; 1999b; Cox, 1996; Komarek et al., 1998; Komarek and Anagnostidis, 1999; Baker and Fabbro, 2002; John et al., 2003; Krammer, 2003; Komárek and Anagnostidis, 2005; Joosten, 2006; Hofmann et al., 2011; Park, 2012; Komárek, 2013; Taşkın et al., 2019). Taxa and author names were identified following standardized databases (Guiry and Guiry, 2022). At least 400 cells were counted for all of the samples (phytoplankton and diatoms).
2.4 Phytoplankton indices and calculate of ecological quality
The ecological quality of the Eastern Mediterranean River basin was assessed using five biotic and five abiotic indices. These indices were calculated to determine the ecological quality of the lakes. Among the biotic indices, the Assemblage Index (Q), Mediterranean Phytoplankton Trophic Index (MedPTI), Phytoplankton Biovolume Index (PT-BV) and Phytoplankton Trophic Index (PTI) were calculated using phytoplankton biovolume, while the relative abundance of diatom species was taken into account in calculating the Trophic Diatom Index for Lakes (TDIL) values. In the Assemblage Index (Q), the phytoplankton species into functional groups representing more than 5% of total biovolume were selected, and the index was estimated by Reynolds et al. (2002) and Padisák et al. (2006). The Mediterranean Phytoplankton Trophic Index (MedPTI) was calculated as trophic values (v) and indicator values (i) by Marchetto et al. (2009). The Trophic Diatom Index for Lakes (TDIL) was calculated applying the equation modified by Zelinka & Marvan, (1961) in the Omnidia v6.0 software (Stenger-Kovacs et al., 2007). The Phytoplankton Biovolume Index (PT-BV) was evaluated according to total biovolume in deep and shallow lakes (DGWM, 2020). The Phytoplankton Trophic Index (PTI) was calculated according to the optimum value of the taxon in the sample (Phillips et al., 2013). Trophic State Index (TSI), Trophic Level Index (TLI), Water Quality Index minimum (WQImin-nw), Water Quality Index (WQI), and Light Attenuation Coefficient (Kna) indices were calculated by environmental water quality parameters as abiotic indices. The trophic condition of the study area was determined using Carlson's Trophic State Index (TSI), and Trophic Level Index (TLI) values (Carlson, 1977; Burns et al., 1999). In addition to trophic assessments, the water quality status of the sampling stations were assessed by Kükrer and Mutlu (2019) and Wu et al. (2021) using Water Quality Index (WQI) and Water Quality Index minimum (WQImin-nw) methods, to more objectively evaluate the water quality status of the study areas and compare them with biotic indices. Also, the computation of the light attenuation coefficient (Kna) was used by Walker (1982) and Nayek et al. (2018) to assess the underwater light availability in the study.
2.5 Statical analysis
Prior to conducting statistical analysis, the datasets containing information on environmental parameters and indices were evaluated for normality and homogeneity through the Kolmogorov-Smirnov test. Non-parametric tests were then used for all mathematical-statistical procedures, as recommended by Nejumal et al. (2021). To measure the correlation between the environmental parameters and indices (both biotic and abiotic), the Spearman correlation matrix was utilized and analyzed with the SPSS v18.0 statistical software designed for Windows (SPSS for Windows, 2008).
Principal component analysis (PCA) was utilized to identify the fourteen water quality parameters that showed significance in the data set. Outliers in relation to these parameters were also identified through the use of PCA. To investigate patterns in phytoplankton assemblages, detrended correspondence analysis (DCA) was employed. The gradient lengths obtained through DCA were used to confirm that unimodal response models were the best method for examining species responses to environmental parameters. Due to the gradient length of axis 1 in DCA analysis being longer than 4, the relationship between environmental parameters and phytoplankton assemblages was determined by Canonical Correspondence Analysis (CCA) using the Canoco v5.0 software (Smilauer, 2012). A Model of the logarithmic transform “Log(X + 1)” for the decrease in skewness was applied for the environmental parameters (except for pH). The CCA analysis was tested in the Monte Carlo simulation (499 unrestricted permutations, p < 0.05) to clarify the significance of the environmental parameters on phytoplankton data (Leps and Smilauer, 2003; Ter Braak CJF, Šmilauer, P. 2012).
3 Results
3.1 Hydrographic parameters
Among the seven monitoring lakes, the highest values of most environmental water quality parameters (temp, pH, EC, SS, COD, BOD5, TN and TP) were measured in Akgöl Lake (AL2) which is a shallow lake, while the lowest values for temp, EC and SS were mostly recorded in Sarıveliler Pond (SL6). The lowest dissolved oxygen value (5.97 mg L−1) was recorded in Akgöl Lake (AL2) during autumn sampling, while Berdan Dam Lake (SL4) had the highest value (9.2 mg L−1) in spring season. The lentic systems in the Eastern Mediterranean River had slightly alkaline waters. The highest mean pH value (8.6) was recorded in station AL2 (Akgöl Lake), and Gezende Dam Lake (AL1) showed the lowest pH level (7.3). Relatively low values of total organic carbon (TOC) were measured in Gezende Dam Lake (AL1), Berdan Dam Lake (AL4, SL4), Ermenek Dam Lake (AL7), and Akgöl Lake (SL2) as the following: 3.9, 3.7, 3.8, 3.7 and 3.9 mg L−1, respectively. The highest mean TOC value (16.5 mg L−1) was recorded in Bağbaşı Dam Lake (AL5). The water temperature showed similar spatio temporal patterns to the EC gradient in the lakes surveyed.
Sampling sites in the Eastern Mediterranean system had a mostly low nutrient gradient except Akgöl station (AL2). The highest mean TP was found in Akgöl Lake (AL2) with 0.14 mg L−1, whereas the lowest value (0.0045 mg L−1) was determined in Alaköprü Dam Lake (AL3), Bağbaşı Dam Lake (AL5), and Sarıveliler Pond (AL6). Station SL6 had also the lowest TN (0.09 mg L−1), temp (5.5 °C), EC (192 μS cm−1), and SS (8.9 mg L−1) values. Alaköprü Dam Lake (AL3) consisted of the highest mean NH4+-N with 0.083 mg L−1. Mainly, low values of dissolved oxygen and high values of chemical oxygen demand and total nitrogen were found in the sampling sites of the Eastern Mediterranean system during autumn sampling period.
3.2 Phytoplankton dynamics
In the Eastern Mediterranean River basin, a total of 90 phytoplankton taxa from eight major taxonomic categories were found. Out of these, 32 taxa were selected for analysis because they constituted more than 1% of the total biovolume and were observed at least three times. The names, codes, and life-forms of these 32 taxa are presented in Table 2. While 46 phytoplankton species were only reported once at the monitoring stations, 44 species were observed more than once in the phytoplankton of the lake systems. During the study, Pantocsekiella ocellata, Dinobryon divergens, Fragilaria tenera var. nanana, and Cryptomonas ovata were frequently found. Miozoa and Bacillariophyta members were the main contributors to the biovolume in the Eastern Mediterranean River basin, mainly represented by Peridinium cinctum, Ceratium hirundinella, and Gyrosigma balticum respectively. While both divisions contributed equally to the total biovolume of the basin in the autumn season, it was seen that the Miozoa members came in to prominence due to individual dominance in the spring season. The total biovolume in the spring period was lower than in the autumn, with the exception of the L1 and L3 sampling sites. Miozoa members contributed equally to the total biovolume of the basin in both sampling periods (autumn and spring seasons). On the other hand, Bacillariophyta was the dominant division of the basin in the autumn period, but it did not maintain this dominance in the spring and its biovolume decreased considerably. While the biovolume distributions of both dominant groups of phytoplankton at the sampling sites in the spring season were balanced, it differed between the lakes in the autumn season. In the autumn season, the phytoplankton biovolume was dominated by diatoms (40.4%), and the greatest contribution to the biovolume came from G. balticum species, which peaked at the Akgöl Lake (AL2). In seven monitoring lakes of the river basin, Miozoa and Euglenozoa members contributed subdominantly to the phytoplankton biovolume in the autumn season (31.9% and 16.2%, respectively). However, dinoflagellates were important contributors to biovolume (64.4%) in the spring season, and Peridinium cinctum and Peridiniopsis kulczynskii from Miozoa peaked in Berdan Dam Lake (SL4). In the phytoplankton of the basin, diatoms had the highest number of species (34 taxa), although they did not contribute as much to the biovolume of the lakes as dinoflagellates, which had only 5 taxa.
List of the 32 taxa with names, codes and life-forms.
3.3 Statistical observations
The Spearman correlation coefficient (SCC) was used to reveal the relationships between parameters and indices in Table 3. The Kolmogorov-Smirnov test showed that all parameters were not normally distributed (p < 0.05) except the pH and DO (p > 0.05). Results of the Spearman correlation analysis revealed that abiotic indices (especially, TSI, TLI, and WQI) have more significant correlations with most indices and parameters than biotic indices (Tab. 3). For instance, WQI, an abiotic index, showed significantly positive correlations with PT-BV, TDIL, TSI, and TLI indices (p < 0.01), while this abiotic index correlated negatively with MedPTI and WQImin-nw (p < 0.01). Among indices, very strong associations were seen between TSI with TLI, and WQI with WQImin-nw (r > 0.8). According to the Spearman correlation between environmental parameters and abiotic indices (Tab. 3), electrical conductivity (EC) and dissolved oxygen (DO) parameters correlated with all abiotic indices, while some water quality parameters such as chemical oxygen demand (COD), temperature (temp), and total nitrogen (TN) showed a relation with most abiotic indices except Kna (p < 0.01). However, ammonium (NH4+) and total organic carbon (TOC) parameters had no association with any abiotic indices (correlation coefficients: ±0.0 to ±0.2). Among the aforementioned parameters, DO, COD and TN showed the best relationship with all indices, and the correlation strengths of the three parameters with the indices were at moderate association (correlation coefficients: ±0.4 to ±0.6). It was observed that there was a positive correlation between WQI and some environmental parameters (temp, EC, COD, TN), however, this abiotic index had a negative correlation with DO parameter (p < 0.01). TSI and TLI had significant positive correlations with temp, pH, EC, COD, and TN parameters, while negatively interacting with DO. Kna, non-algal light attenuation, was negatively correlated with DO (p < 0.05) and positively correlated with EC, SO4 (p < 0.01) and TP (p < 0.05). The correlation between environmental parameters and biotic indices are shown in Table 3. DO, COD and TN parameters indicated significant positive and negative correlations with most biotic indices (p < 0.01). Among the biotic indices, TDIL index showed the significant correlations with parameters (p < 0.01), while PTI index did not have significant associations with water quality parameters (p > 0.05). Although Q index, MedPTI, and TDIL biotic indices correlated negatively with most environmental parameters, PT-BV index correlated positively with parameters such as temp, EC, SS, SO4, COD, TN, COD and TN parameters indicated a strong correlation with TDIL (r > 0.6), while temp, EC, SS, TOC, SO4, BOD5, COD, and TP parameters displayed very weak association with this biotic index (r < 0.2).
The Principal Component Analysis (PCA) was used to reveal the relationships between environmental parameters (arrow) and sampling sites (circle) in Figure 2. The first two PCA axes accounted for 95.4% of the total variability as explained in the PCA plot pattern. The PCA ordination showed a good relationship between lakes and environmental parameters as reflected by the high lake-environment correlation coefficient associated with each axis. L3 and L5 sampling sites (lakes) were grouped closely and were associated with high total organic carbon and ammonium concentrations. L7 was associated with high dissolved oxygen concentrations. L2 sampling site was linked to high SS, EC, TP, temp and BOD5 values as also demonstrated by the significant correlations between them in the biplot ordination. Furthermore, a positive relationship detected between L4 and high concentrations of some parameters (SO4, NO3, TN), and these parameters were opposed to the L3 and L5 sampling sites in the PCA ordination (Fig. 2).
For the whole sampling set of lake systems, the eigenvalues of the first two CCA axes (λ1 = 0.729, 27.8% of variance explained and λ2 = 0.505, 47.1% of variance explained) were both significant (p < 0.05; Monte Carlo permutation test, 499 random permutations), and they explained 91.6% of the total variation (2.62) in the species data. The species-parameter correlations for CCA axis 1 (0.99) and 2 (0.96) were high, indicating a relatively strong relationship between phytoplankton assemblages and environmental parameters. Of the phytoplankton taxa, 32 species with a biovolume greater than 1% were used in multivariate statistical analysis (CCA).
After a forward selection 14 environmental water quality parameters formed the best set of parameters that explained a statistically significant amount of the total variation in the phytoplankton communities (Fig. 3). A comprehensive overview of Canonical Correspondence Analysis, electrical conductivity (EC), suspended solids (SS), total phosphorus (TP), biological oxygen demand (BOD5), nitrite (NO2-N), ammonium (NH4+-N), and total nitrogen (TN) were the environmental parameters that accounted for significant portions of the total variance in phytoplankton species composition (p < 0.05). EC, TP, SS, BOD5 parameters (explaining 23.8, 23.7, 22.5 and 19.7% of the total variance in the phytoplankton assemblages, respectively) showed high positive correlations with the first CCA axis and negative correlations with NO2-N (explaining 15.2% of the variance). The second CCA axis indicated a positive correlation with ammonium (NH4+-N) (explaining 15.6% of the variance). As seen in the CCA ordination diagram, pollution-tolerant species such as Cocconeis placentula var. euglypta, Gyrosigma balticum, Euglena repulsans, E. granulata, Lepocinclis oxyuris, L. acus, and Trachelomonas hispida taxa were located on the positive side of axis 1 associating especially with SS, EC, temp, and TP parameters. Another phytoplankton species situated on the upper right side of the biplot was the pollution-tolerant Euglenaformis proxima species, which displayed a positive correlation with TN. However, pollution-sensitive organisms (e.g., Fragilaria tenera var. nanana, Gyrosigma attenuatum, Pantocsekiella ocellata, Peridinium cinctum, Peridiniopsis kulczynskii, P. quadridens) and facultative species (Nitzschia sigmoidea, Euglena velata, Ceratium hirundinella, Cryptomonas ovata, Gyrosigma acuminatum) were located on the lower negative side of axis 1 associating with DO. Oocystis lacustris and Staurastrum gracile species exhibited a close relationship with ammonium (NH4+-N) on vertical axis. Nitrite (NO2-N), another nitrogenous nutrient, displayed association with facultative Ulnaria ulna and Dinobryon divergens species on the upper left side of the plot.
Spearman rank correlation between indices and environmental parameters (N = 87). The meanings of the coefficients in the table is as follows: very weak or no association (±0.0 to ±0.2), weak association (±0.2 to ±0.4), moderate association (±0.4 to ±0.6), strong association (±0.6 to ±0.8), very strong association (±0.8 to ±1.0).
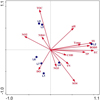 |
Fig. 2 PCA diagram of sampling sites (circle) and environmental parameters (arrow) relationships. Water temperature (Temp), electrical conductivity (EC), suspended solids (SS), ammonium (NH4+), nitrite (NO2), nitrate (NO3), total nitrogen (TN), sulfate (SO4), total phosphorus (TP), dissolved oxygen (DO), biological oxygen demand (BOD5), chemical oxygen demand (COD), total organic carbon (TOC), and potential hydrogen (pH). The full names and abbreviation codes of the lakes are given in Table 1. |
 |
Fig. 3 CCA diagram of species (circle) and environmental parameters (arrow) relationships. Water temperature (Temp), electrical conductivity (EC), suspended solids (SS), ammonium (NH4+), nitrite (NO2), nitrate (NO3), total nitrogen (TN), sulfate (SO4), total phosphorus (TP), dissolved oxygen (DO), biological oxygen demand (BOD5), chemical oxygen demand (COD), total organic carbon (TOC), and potential of hydrogen (pH). The full names abbreviation codes of 32 taxa are given in Table 2. |
3.4 Ecological status of the lakes
Table 4 presents the bioassessment results of the sampling sites based on different biotic and abiotic indices. These indices, developed in various regions, indicated different ecological statuses for the Eastern Mediterranean Lake Systems, ranging from high to poor conditions. Among the abiotic indices, WQImin-nw displayed the largest variation in ecological status between lakes and seasons. On the other hand, MedPTI could not distinguish the ecological status of the stations and seasons, except for AL2, but it indicated a high ecological status for all stations.
The ecological results of TSI and TLI indices for all stations were exactly similar. Q index, TDIL, PTI, WQI, and Kna indices made a small distinction in the environmental conditions of the sampling sites. Deterioration of AL2 station was represented by several indices, which indicated a poor ecological status based on PT-BV, PTI, TSI, TLI, WQI, WQImin-nw and Kna; MedPTI indicated a bad ecological status. However, results of Q index and TDIL indicated that AL2 station had a high and moderate environmental condition, respectively. Q index, MedPTI, TSI, and TLI indices showed mainly similar behavior to differentiate the environmental conditions of the sampling sites in the Eastern Mediterranean River basin. Results of these indices indicated that AL1, AL3, AL4, AL5, AL6, AL7, SL1, SL2, SL3, SL4, SL5, SL6, and SL7 sampling sites were mostly in high ecological status. However, WQI and Kna from abiotic indices and TDIL from biotic indices indicated that most lake systems had good ecological conditions, which is a subclassification of high quality (Tab. 4).
Ecological status of the seven sites (lakes) according to various biotic and abiotic indices. The meaning of different colors used in the table is as follows: blue − high quality, green − good quality, yellow − moderate quality, orange − poor quality, red − bad quality.
4 Discussion
The study identified key indices that most accurately reflect the ecological status of the lakes based on environmental variables and phytoplankton communities. Indices highlighted the significant effect of phytoplankton communities and physico-chemical variables on lake systems. Phytoplankton community structure and parameters in this study closely followed the observed changes in pollution levels as a result of changes in the lake systems. The index results indicated that the highly polluted sampling site, specifically Akgöl Lake, showed notable differences in phytoplankton communities compared to the less polluted sampling sites within the basin. The algal communities were primarily affected by inorganic dissolved solids (as conductivity) and nutrient concentration (amonium) in the lakes resulting from agricultural runoff and climatic environment. Specifically, conductivity had a greater impact on shallow lake (as Akgöl Lake), while ammonium played a more significant role in other lakes located in distal areas. The prominence of EC in Akgöl Lake suggests that the mineral content and salinity of the water play a significant role in determining its water quality. Conversely, the importance of ammonium in the other oligo-mesotrophic lakes suggests that nitrogen compounds, specifically in the form of ammonium, is a crucial factor in assessing their water quality. Oligo-mesotrophic lakes typically have lower nutrient levels and are less productive compared to meso-eutrophic lakes.
Based on PCA analysis, EC, SS, BOD5, temp, DO, TOC, TP, SO4, NH4+, NO3, and TN parameters exhibited a positive relationship with each other and played a significant role in shaping the water quality characteristics of the lakes (p < 0.05). Their strong influence on the principal components suggests that changes in these parameters are likely to reflect variations in overall water quality. However, certain parameters such as pH, NO2, and COD had a lesser influence on the principal components or showed an inverse relationship with the other variables in the analysis. This implies that these parameters may not play a major role in determining the overall water quality in the lakes studied. The ecological status of the seven lakes in the Eastern Mediterranean River basin was stratified into two types on the basis of some environmental variables that have a strong influence on lake's algal community and productivity. Meso-eutrophic status falls into the first type, characterized by Akgöl Lake is primarily responsible for its low water quality classification, causing an increase in more pollution-tolerant species in this water body. The second type, oligo-mesotrophic structure, was characterized by eight other lakes in the basin. Unlike the first type, these lakes have lower levels of nutrients and are considered to have better water quality. Regarding the specific sampling sites within the lake basin, the PCA results indicated that the L2 sampling site exhibited the closest relationship with the water quality parameters (EC, SS, BOD5, temperature, and TP) among the seven lakes. This suggests that these environmental parameters measured at L2 were characteristic of that specific lake and can be considered representative of its water quality. These findings align with previous studies conducted by Ersanlı (2001), Pereira et al. (2005), Soylu (2006), Alp et al. (2016), Mangadze et al. (2017) and Balasubramaniam et al. (2018), which have demonstrated the influence of the above-mentioned parameters on algal abundance and distribution. However, TP and PO4-P parameters were found to be effective on the abundance and distribution of algae in shallow lakes with low conductivity such as Küçük Akgöl Lake (Ongun-Sevindik et al., 2023) and Ladik Lake (Maraşlıoğlu et al., 2005). Furthermore, the PCA analysis showed that the L1 and L6 sampling sites did not directly correlate with any environmental parameter in the ordination, unlike other lakes in the basin. This suggests that the water quality parameters measured at L1 and L6 may be influenced by different underlying factors not captured in the study. It is possible that other variables or local conditions, not considered in the analysis, are driving the variations in water quality at those particular sites.
Gyrosigma balticum is a pollution-tolerant species that has contributed significantly to the total biovolume in a study. The presence of undamaged G. balticum cells in water samples from Akgöl lake with high salinity indicated that this species is typically found in estuarine environments characterized by high conductivity levels. Its abundance and contribution to the total biovolume indicate its ecological significance in polluted aquatic ecosystems. The observation aligns with previous research studies (Bere and Tundisi, 2011; Sahu et al., 2012; Pednekar et al., 2014; Balasubramaniam et al., 2018), which demonstrated a correlation between high conductivity levels and increased diatom diversity. Additionally, G. balticum exhibited a peak biovolume during autumn sampling in Akgöl Lake, coinciding with high suspended solids levels, and similar patterns have been observed in other study from different location (Pereira et al., 2005). The similarity of these findings with the study conducted in Olinda-PE (Brazil), reinforces the notion that G. balticum's response to environmental stressors, such as high suspended solids, is consistent across different locations. The presence of this diatom species in significant quantities during periods of high suspended solids may indicate the species' ability to adapt and thrive in these stressed environments. The higher total phosphorus concentration observed during the G. balticum bloom in autumn supports the notion that phosphorus availability influenced the growth and abundance of this diatom species. The lower phosphorus concentration during spring, when G. balticum was absent, indicates that the absence of this species may be attributed to the limited phosphorus availability during that period. This result align with the finding reported by Pereira et al. (2005), emphasizing the importance of phosphorus in the dynamics of G. balticum. The overlapping finding from different study suggest a consistent pattern and reinforce the significance of phosphorus as a limiting factor in the growth and proliferation of this species.
The data provided by CCA analysis suggest that there is a negative correlation between P. cinctum, a freshwater dinoflagellate, and ammonium levels (p < 0.05). This finding is in agreement with previous studies conducted by Sanz-Luque et al. (2015), Öterler (2018), and Darki and Krakhmalnyi (2019), which also reported that low nitrogen levels stimulate the growth of P. cinctum cells. The predominance of P. cinctum in lightly polluted freshwater ecosystems, as observed in studies by Starmach (1974), Padisak (1985), Grigorszky et al. (2006) and Öterler (2018), further supports its association with such environments. It is considered an indicator species associated with low trophic levels, as indicated by Reynolds et al. (2002) and Darki and Krakhmalnyi (2019). Furthermore, P. cinctum species has a widespread distribution in waters with low-organic compounds, particularly in the Eastern Mediterranean basin with an oligo-mesotropic character, as exemplified by the Berdan Dam Lake.
The Spearman's correlation analysis revealed that the abiotic indices showed more significant correlations both with each other and with environmental parameters than with biotic indices. The similarity of some environmental parameters used in the calculation of abiotic indices such as TSI and TLI, and also WQI and WQImin-nw caused a strong correlation between these indices. The biotic indices generally didn't show significant relationships with each other. The strongest correlation among biotic indices was found between Q index and TDIL (r = –0.578, p < 0.01). Although both indices are used for shallow lakes, the fact that the TDIL is diatom-based and the Q index is phytoplankton-based makes the negative correlation between the two indices meaningful. The fact that most of the lakes in the study basin are not shallow lakes has caused this contrast correlation for Q index which was specially developed for Hungarian shallow lakes was found more convenient in Hungarian lakes such as Lake Balaton (Bolla et al., 2010) than other European lakes such as Great Lake in Albania (Vidaković et al., 2020) and French Atlantic lakes (Cellamare et al., 2012). Therefore, the TDIL from biotic indices seems to be a promising tool for ecological status evaluation of lakes in the Eastern Mediterranean system.
Based on the Spearman's correlation results between the indices and the environmental variables, it was found that TSI, TLI and WQI abiotic measurements showed strong positive correlations with parameters such as temp, EC, DO, COD, NO2, TN, and TP. Similarly, high positive correlation (R2 = 0.78) was observed between WQI and DO in Siling Reservoir (Hashmi et al., 2016). Additionally, the level of DO variability was strongly correlated with TSI (p < 0.05) in six lakes located in five different districts in urban Hanoi (Viet et al., 2016). Among the biotic indices, the strong correlations oberved between the TDIL and parameters such as COD and TN suggest that the diatom communities assessed by the TDIL index are particularly responsive to variations in COD-induced organic pollution and TN-induced nutrient enrichment in aquatic environments (r > 0.6, p < 0.01). However, it is worth noting that most of the Hungarian lakes classified as being in bad or poor status according to the TDIL index are characterized as shallow, saline lakes with naturally high TP content and high conductivity (Stenger-Kovács et al., 2007). It is important to consider these limitations and contextual factors when applying biotic indices like the TDIL index. Considering the above information, it can be concluded that various oxidizable pollutants (such as COD) and nutrients (such as TN and TP) have a significant influence on the indicator diatom communities used in the calculation of the TDIL index. The pollution tolerance index, another biotic index commonly used in lakes, classified all lakes in a lower class (mesotrophic) compared to other indices that indicated good to excellent water quality in the Eastern Mediterranean basin. The negative correlation observed between PTI and DO levels in L2 sampling site implies that when the dissolved oxygen level in a lake is low, it indicates poorer water quality, which is associated with a high PTI value. On the other hand, in other lakes within the Eastern Mediterranean systems where oxygen levels are higher, the water quality conditions are better, and this is associated with relatively lower PTI values. The studies by Çelekli et al. (2018) and Çelekli and Özpınar (2021) also support the existence of this negative relationship between PTI and DO levels. Additionally, it was found that the correlation between the PTI and TP was very weak (r = 0.004, p > 0.05) due to the low average TP value in the studied lake systems (0.03 mg L−1). Accordingly, when TP levels are low in a lake, the PTI index may not adequately differentiate between different ecological statuses or effectively reflect water quality variations. The PTI index, which has been found to be strongly correlated with TP in previous studies conducted in European freshwater bodies (Philips et al., 2013; Çelekli et al., 2018; Çelekli and Özpınar (2021)), may not provide sufficient discrimination among ecological statuses or accurately reflect water quality conditions in systems with low TP loads like our study area. For this reason, PTI is well suited for environments with higher TP levels and more pronounced eutrophication pressures.
Differences in species diversity and environmental parameters between sampling sites reflected on all indices except PTI. Most of these indices captured and integrated the effects of species diversity and various environmental conditions on the overall ecological status of the lakes. Otherwise, the PTI, which specifically focuses on phosphorus concentrations, was not be as sensitive to variations in species diversity and certain environmental parameters. Based on the average values and monthly changes of all indices values during the studied period, it was found that the diatom-based TDIL and the physicochemical-based WQI indices were more representative indices of the ecological status of lakes in the Eastern Mediterranean system. Both indices classified the ecological status of the majority of the sampling sites in the basin as good quality, except for one site (L2). These indices provided a holistic view of ecosystem health, taking into account both biological and physicochemical aspects of water quality.
Assessing the ecological status of lentic ecosystems requires the development of integrated approaches that consider the complex relationships between bio-indicator assemblages and ecological factors. Since the application of the European WFD (EC, 2000), the biological assessment of surface water quality has been a more important issue to accomplish environmental sustainability. The evaluation results of the sampling sites indicated that the indices developed from biotic-based and abiotic-based different parameters have different scores resulting in different ecological status from high to bad condition in the Eastern Mediterranean River systems. In the ecological evaluation of the sampling sites, PTI from biotic and WQImin-nw from abiotic reflect partially low ecological status, while MedPTI from biotic and TSI, TLI from abiotic have high ecological status. Biotic indices such as Q index, PT-BV, MedPTI, and TDIL mostly showed similar behavior in the bioassessment of ecological conditions of sampling sites in the Eastern Mediterranean basin. Similar findings for TDIL and Q indices were also detected in two shallow Mediterranean lakes (Taşkısığı Lake and Little Akgöl Lake) in Sakarya river bed (Sevindik et al., 2017; Ongun-Sevindik et al., 2023). The PTI index, on the other hand, differed partially from other biotic indices and showed moderate conditions in the basin due to having the different tolerance/sensitivity values of phytoplankton species. The all indices exposed that the water quality of the lentic systems in the Eastern Mediterranean region was better during spring and Akgöl was a shallow lake with the lowest ecological status among the seven lakes in the Eastern Mediterranean River basin. Some phytoplankton species and environmental parameters used in the calculation of biotic and abiotic indices played a significant role on the improving the ecological status in the lakes during spring season. The assessment using some biotic and abiotic indices gave compatible results for determining the ecological status of lake systems in in the Eastern Mediterranean River basin.
5 Conclusion
The present study pointed out that certain phytoplankton species and environmental parameters used in the calculation of biotic and abiotic indices have a significant impact on improving the water quality of the lakes, particularly during the spring season. The evaluation results of the sampling sites in the Eastern Mediterranean River systems indicated that the available indices developed from biotic-based and abiotic-based parameters, leading to different ecological status classifications. These classifications can range from a high ecological condition to a bad ecological condition, indicating variations in water quality among the different sampling sites. The results for all available indices consistently indicated that the distal sites (L1, L3, L4, L5, L6, and L7) have a high ecological status. This suggested that these sites generally exhibit better water quality and are considered to be in good ecological condition compared to the other sampling site (L2). According to the present study, it appears that the diatom-based TDIL and the physicochemical-based WQI could be more suitable indices for assessing the ecological status of lentic systems in the Mediterranean region. Both indices provided a more comprehensive approach to water quality assessment of lakes in the basin.
The results of the present study revealed that electrical conductivity (EC) was identified as the main environmental water quality parameter in the meso-eutrophic Akgöl Lake, whereas in the six oligo-mesotrophic lakes, nitrogen-based ammonium (NH4+) was found to be a more successful parameter in assessing water quality. However, it is important to note that the statistical analysis findings of the study support the notion that no single environmental parameter has a limiting effect on species diversity and algal biovolume. This means that the ecological status of the lakes is determined by multiple interacting factors, both biotic and abiotic. While EC and NH4+ may be key indicators in assessing water quality in the seven lakes of the basin, other parameters and biotic factors also play significant roles in determining the ecological status of the lakes.
Consequently, it would be more accurate to use biotic indices based on diatom or phytoplankton and abiotic indices based on physicochemical parameters together in determining the ecological status of lentic systems. Thus, by integrating abiotic data with biological assessments, a more comphrehensive understanding of water quality and ecosystem health can be obtained. This integration allows for a multidimensional assessment that considers the interactions and feedbacks between the biological and environmental components of the ecosystem.
References
- Abboud-Abi Saab M. 1992. Day-to-day variation in phytoplankton assemblages during spring blooming in a fixed station along the Labanese coastline. J Plankton Res 14: 1099–1115. [CrossRef] [Google Scholar]
- Alp M, Fakioglu Y, Ozbay O, Kocer M. 2016. A study on water quality and trophic state of Akgol Lagoon (Mersin, Turkey). Aquat Ecosyst Health Manag 19: 58–63. [CrossRef] [Google Scholar]
- APHA. 2012. American Water Works Association, Water Environment Federation, Standard Methods for the Examination of Water and Wastewater. 22nd Edition, 1496p. [Google Scholar]
- Baker PD, Fabbro LD. 2002. A guide to identification of common blue-green algae (cyanoprokaryotes) in Australian freshwaters. Co-operative Research Centre for Freshwater Ecology Identification, Vol. 25, Thurgoona, Australia. [Google Scholar]
- Balasubramaniam J, Marimuthu P, Jayaraj KA. 2018. Two new record of benthic diatom − Diploneis crabro (Ehrenberg) and Gyrosigma balticum (Ehrenberg) Rabenhorst from the mangrove intertidal regions of South Andaman, Andaman archipelago, India. Indian J Geo-Mar Sci 47: 1237–1239. [Google Scholar]
- Bayer-Altin T, Altin BN. 2021. Response of hydrological drought to meteorological drought in the Eastern Mediterranean Basin of Turkey. J Arid Land 13: 470–486. [CrossRef] [Google Scholar]
- Becker V, Huszar VLM, Naselli-Flores L, Padisák J. 2008. Phytoplankton equilibrium phases during thermal stratification in a deep subtropical reservoir. Freshw Biol 53: 952–963. [CrossRef] [Google Scholar]
- Bellinger EG, Sigee DC. 2010. Freshwater algae: Identification and use as bioindicators. Wiley-Blackwell: Chichester, West Sussex, UK. [CrossRef] [Google Scholar]
- Bennion H, Kelly MG, Juggins S, Yallop ML, Burgess A, Jamieson BJ, Krokowski J. 2014. Assessment of ecological status in UK lakes using benthic diatoms. Freshw Sci 33: 639–654. [CrossRef] [Google Scholar]
- Bere T, Tundisi JG. 2011. Influence of ionic strength and conductivity on benthic diatom communities in a tropical river (Monjolinho), São Carlos-SP, Brazil. Hydrobiologia 661: 261–276. [Google Scholar]
- Borics G, Tóthmérész B, Lukács BA, Várbíró G. 2012. Functional groups of phytoplankton shaping diversity of shallow lake ecosystems. Hydrobiologia 698: 251–262. [CrossRef] [Google Scholar]
- Bortolini JC, Rodrigues LC, Jati S, Train S. 2014. Phytoplankton functional and morphological groups as indicators of environmental variability in a lateral channel of the Upper Paraná River floodplain. Acta Limnolog Bras 26: 98–108. [CrossRef] [Google Scholar]
- Burns NM, Rutherford JC, Clayton JS. 1999. A monitoring and classification system for New Zealand Lakes and reservoirs. Lake Reserv Manag 15: 255–271. [CrossRef] [Google Scholar]
- Caballero M, Rodriguez A, Vilaclara G,Ortega B, Roy P, Lozano García S. 2013. Hydrochemistry, ostracods and diatoms in a deep, tropical, crater lake in Western Mexico. J Limnol 72: 512–523. [Google Scholar]
- Carlson RE. 1977. A Trophic State Index for Lakes. Limnol Oceanogr 22: 361–369. [CrossRef] [Google Scholar]
- Carvalho L, Poikane S, Lyche Solheim A,Phillips G, Borics G, Catalan J, Thackeray SJ. 2013. Strength and uncertainty of phytoplankton metrics for assessing eutrophication impacts in lakes. Hydrobiologia 704: 127–140. [CrossRef] [Google Scholar]
- Cellamare M, Morin S, Coste M, Haury J. 2012. Ecological assessment of French Atlantic lakes based on phytoplankton, phytobenthos and macrophytes. Environ Monit Assess 18: 4685–4708. [CrossRef] [PubMed] [Google Scholar]
- Çelekli A, Özpınar G. 2021. Ecological assessment of Burç Reservoir's surface water (Turkey) using phytoplanktonmetrics and multivariate approach. Turk J Botany 45: 522–539. [Google Scholar]
- Çelekli A, Kayhan S, Çetin T. 2020. First assessment of lakes' water quality in Aras River catchment (Turkey); Application of phytoplankton metrics and multivariate approach. Ecol Indic 117: 106706. [CrossRef] [Google Scholar]
- Çelekli A, Toudjani AA, Lekesiz HÖ, Kayhan S, Çetin T. 2018. Bioassessment of ecological status of three Aegean reservoirs based on phytoplankton metrics. Turk J Water Sci Manag 2: 76–99. [CrossRef] [Google Scholar]
- Chapman AD, Pfiester LA. 1995. The effect of temperature, irradiance, and nitrogen on encystment and growth of the freshwater Dinoflagellates Peridinium cinctum and P. willei in culture (Dinophyceae). J Phycol 31: 355–359. [CrossRef] [Google Scholar]
- Cox EJ. 1996. Identification of Freshwater Diatoms from Live Material. London: Chapman and Hall, pp. 1–158. [Google Scholar]
- Danladi Bello AA, Hashim NB, Haniffah MRM. 2017. Predicting impact of climate change on water temperature and dissolved oxygen in tropical rivers. Climate 5: 58p. [CrossRef] [Google Scholar]
- Darki BZ, Krakhmalnyi AF. 2019. Biotic and abiotic factors affecting the population dynamics of Ceratium hirundinella, Peridinium cinctum, and Peridiniopsis elpatiewskyi. Diversity 11: 137–149. [Google Scholar]
- Demir AN, Fakıoğlu Ö, Dural B. 2014. Phytoplankton functional groups provide a quality assessment method by the Q assemblage index in Lake Mogan (Turkey). Turk J Bot 38: 169–179. [CrossRef] [Google Scholar]
- DGWM (Ministry of Agriculture and Forestry, GeneralDirectorate of Water Management). 2013. Project on preparation of basin protection action plans in Eastern Mediterranean Basin. Directorate General of Water Management, Ankara, Türkiye. [Google Scholar]
- DGWM (Ministry of Agriculture and Forestry, GeneralDirectorate of Water Management). 2015. Türkiye'de Havza Bazında Hassas Alanların ve Su Kalitesi Hedeflerinin Belirlenmesi Projesi. Final Raporu, Cilt- 1. [Google Scholar]
- DGWM (Ministry of Agriculture and Forestry, GeneralDirectorate of Water Management). 2016.Climate change impacts on water resources project. Eastern Mediterranean Region, Project No. 19. Ankara: Republic of Turkey Ministry of Agriculture and Forestry General Directorateof Water Management Publishing, 1–140 pp. [Google Scholar]
- DGWM (Ministry of Agriculture and Forestry, GeneralDirectorate of Water Management). 2020. Project fort the Establishment of a Reference Monitoring Network in Türkiye. Final Report. Directorate General of Water Management, Ankara, Türkiye. [Google Scholar]
- EC. 2000. European Committee for Standardization, Directive 2000/60/EC of the European Parliament and of the Council of 23 October 2000 establishing a framework for Community action in the field of water policy. Off J Eur Commun Ser L 327: 1–73. [Google Scholar]
- EC. 2014. European Committee for Standardization, Water quality-guidance standard forthe identification and enumeration of benthic diatoms from rivers and lakes. European Standard EN 14407, Brussels. [Google Scholar]
- EEA. 2018. European Waters Assessment of Status and Pressures. EEA Report No 7/2018. European Environment Agency, Luxembourg. [Google Scholar]
- Ersanlı E. 2001. An investigation on algae Lake Simenit (Terme, Samsun-Turkey). Ondokuzmayıs University, Institute of Science, master's thesis no: 105756. 74 p. [Google Scholar]
- Fakıoğlu YE. 2014. Determination of Algal Flora and Water Quality of Akgöl Lagoon (Silifke/Mersin). Mersin University, Institute of Science, master's thesis no: 374401. 127 p. [Google Scholar]
- Gould RW, Balmori JER, Fryxell GA. 1986. Multivariate statistics applied to phytoplankton data from two Gulf Stream warm core rings. Limn Oceanogr 31: 951–968. [CrossRef] [Google Scholar]
- Grigorszky I, Kiss KT, Béres V. 2006. The effects of temperature, nitrogen, and phosphorus on the encystment of Peridinium cinctum, Stein (Dinophyta). Hydrobiologia 563: 527–535. [CrossRef] [Google Scholar]
- Guiry MD, Guiry GM. 2022. AlgaeBase. Galway, Ireland: National University of Ireland. Available at http://www.algaebase.org (accessed: 20 March 2022). [Google Scholar]
- Hashmi MZ, Yu C, Shen C, Muhammad N, Shen H, Chen Y. 2016. Water quality characterization of the siling reservoir (Zhejiang, China) using water quality ındex. Clean Soil Air Water 44: 553–562. [CrossRef] [Google Scholar]
- Hillebrand H, Dürselen CD, Kirschtel D, Pollingher U, Zohary T. 1999. Biovolume calculation for pelagic and benthic microalgae. J Phycol 35: 403–424. [CrossRef] [Google Scholar]
- Hofmann G, Werum M, Lange-Bertalot H. 2011. Diatomeen im Süßwasser-Benthos von Mitteleuropa. Bestimmungsflora Kieselalgen für die ökologische Praxis. Über 700: 908. [Google Scholar]
- Inkel CEV, Hohls BC, Maak EV. 2001. A Ceratium hirundinella (OF Müller) bloom in Hartbeespoort Dam. Water SA 27: 269–276. [Google Scholar]
- Jarosiewicz A, Ficek D, Zapadka T. 2012. Eutrophication parameters and Carlson-type trophic state indices in selected Pomeranian lakes. Limnolog Rev 11: 15–23. [Google Scholar]
- Jekatierynczuk-Rudczyk E, Zieliński P, Grabowska M, Ejsmont-Karabin J, Karpowicz M, Więcko A. 2014. The trophic status of Suwałki Landscape Park lakes based on selected parameters (NE Poland). Environ Monitor Assess 186: 5101–5121. [CrossRef] [PubMed] [Google Scholar]
- John DM, Whitton BA, Brook AJ. 2003. The Freshwater Algal Flora of the British Isles: An Identification Guide to Freshwater and Terrestrial Algae. The Natural History Museum and The British Phycological Society. Cambridge: Cambridge University Press, 700 p. [Google Scholar]
- Joosten AM. 2006. Flora of the Blue-green Algae of the Netherlands: The non-filamentous species of inland waters. Netherlands: KNNV Uitgeverij. [Google Scholar]
- Kamberović J, Stuhli V, Lukıć Z, Habibović M, Mešikić E. 2019. Epiphytic diatoms as bioindicators of trophic status of lake modrac (Bosnia And Herzegovina). Turk J Botany 43: 420–430. [CrossRef] [Google Scholar]
- Kelly M, Juggins S, Guthrie R, Pritchard S, Jamieson J, Rippey B, Hirst H, Yallop M. 2008. Assessment of ecological status in UK rivers using diatoms. Freshw Biol 53: 403–422. [Google Scholar]
- Koçer MAT, Şen B. 2014. Some factors affecting the abundance of phytoplankton in an unproductive alkaline lake (Lake Hazar, Turkey). Turk J Botany 38: 790–799. [CrossRef] [Google Scholar]
- Komárek J, Anagnostidis K. 2005. Süßwasserflora von Mitteleuropa, bd. 19/2: Cyanoprokaryota: Oscillatoriales. Spektrum Akademischer Verlag 19: 1–759. [Google Scholar]
- Komárek J, Anagnostidis K. 1999. Cyanoprokaryota, Vol 19/1: Chroococcales, in Freshwater Flora of Central Europe, edited by H. Ettl, J. Gerloff, H. Heynig, D. Mollenhaueur. Berlin, Heidelberg: Spektrum Akademischer Verlag, 1–548p. [Google Scholar]
- Komárek J. 2013. Cyanoprokaryota, Vol 19/3: Heterocytous Genera, in Freshwater Flora of Central Europe, edited by B. Bübel, G. Gärtner, L. Krienitz, M. Schagerl. Berlin, Heidelberg: Spektrum Akademischer Verlag, pp. 1–1131. [Google Scholar]
- Komarek J, Eloranta P, Lhotski P. 1998. Cyanophyta/Cyanophyta-14. Symposium Internat. Assoc. for CyanophyteResearch (IAC), Lammi (Finland) 1998 / Proceedings, Morphology, taxonomy, ecology. Archiv fur Hydrobiologie (Suppl. 129), Algological studies, 94. [Google Scholar]
- Koyuncu N, Çevik F. 2014. Berdan Baraj Gölü (Mersin) fitoplankton kompozisyonu ve ekolojisi. Ç.Ü. Fen ve Mühendislik Bilimleri Dergisi, 1–31. [Google Scholar]
- Krammer K, Lange-Bertalot H. 1991a. Freshwater flora of Central Europe. Bacillariophyceae, Part 3. Centrales, Fragillariaceae, Eunotiaceae. Germany, Stuttgart. Gustav Fischer Verlag. 576 p. [Google Scholar]
- Krammer K, Lange-Bertalot H. 1991b. Freshwater flora of Central Europe. Bacillariophyceae, Part 4. Achnanthaceae, Critical additions to Navicula (Lineolate) and Gomphonema complete literature. Germany, Stuttgart. Gustav Fischer Verlag. 436p. [Google Scholar]
- Krammer K, Lange-Bertalot H. 1999a. Freshwater flora of Central Europe. Bacillariophyceae, Part 1. Naviculaceae. Germany, Berlin. Spectrum Academicher Verlag. 876 p. [Google Scholar]
- Krammer K, Lange-Bertalot H. 1999b. Freshwater flora of Central Europe. Bacillariophyceae, Part 2. Bacillariaceae, Epithemiaceae, Surirellaceae. Germany, Berlin. Spectrum Academicher Verlag. 610 p. [Google Scholar]
- Krammer K. 2003. Diatoms of Europe, Volume 4, A.R.G. Gantner Verlag K.G. 530 p. [Google Scholar]
- Kükrer S, Mutlu E. 2019. Assessment of surface water quality using water quality index and multivariate statistical analyses in Saraydüzü Dam Lake, Turkey. Environ Monitor Assess 191: 1–16. [CrossRef] [Google Scholar]
- Kutlu B, Serdar O, Aydın R, Danabaş D. 2017. Determination of trophic status Carlson index induced on Uzunçayır Dam Lake (Tunceli). Aquac Stud 17: 83–92. [Google Scholar]
- Leps J, Smilauer P. 2003. Multivariate Analysis of Ecological Data Using CANOCO. Cambridge, UK: Cambridge University Press, 269 p. [Google Scholar]
- Lira GAST, Araujo EL, Bittencourt-Oliveira MDC, Moura AN. 2011. Phytoplankton abundance, dominance and coexistence in an eutrophic reservoir in the state of Pernambuco, Northeast Brazil. Anais da Acad Bras Ciências 83: 1313–1326. [Google Scholar]
- Liu G, Pei G, Hu Z. 2008. Peridiniopsis niei sp. nov. (Dinophyceae), a new species of freshwater red tide dinoflagellates from China. Nova Hedwigia 87: 487–499. [CrossRef] [Google Scholar]
- Lyche-Solheim A, Feld CK, Birk S, Phillips G, Carvalho L, Morabito G, Poikane S. 2013. Ecological status assessment of European lakes: a comparison of metrics for phytoplankton, macrophytes, benthic invertebrates and fish. Hydrobiologia 704: 57–74. [CrossRef] [Google Scholar]
- Mangadze T, Wasserman RJ, Dalu T. 2017. Use of diatom communities as ındicators of conductivity and ıonic composition in a small austral temperate river system. Water Air Soil Pollut 228: 428. [CrossRef] [Google Scholar]
- Maraşlıoğlu F, Soylu EN, Gönülol A. 2005. Seasonal variation of the phytoplankton of lake Ladik Samsun, Turkey J Freshw Ecol 20: 549–553. [CrossRef] [Google Scholar]
- Marchetto A, Padedda BM, Mariani MA, Luglie A, Sechi N. 2009. A numerical index for evaluating phytoplankton response to changes in nutrient levels in deep Mediterranean reservoirs. J Limnol 68: 106–21. [CrossRef] [Google Scholar]
- Margalef R. 1978. Life-forms of phytoplankton as survival alternatives in an unstable environment. Oceanolog Acta 1: 493–509. [Google Scholar]
- Mihaljević M, Špoljarić D, Stević F, Žuna Pfeiffer T. 2013. Assessment of flood-induced changes of phytoplankton along a river-floodplain system using the morpho-functional approach. Environ Monitor Assess 185: 8601–8619. [CrossRef] [PubMed] [Google Scholar]
- Molina-Navarro E, Mart ́ınez-Perez S,Sastre-Merlin A, Verdugo-Althofer M, Padisak J. 2014. Phytoplankton andsuitability of derived metrics for assessing the ecological status in a limno-reservoir, a Water Framework Directivenondefined type of Mediterranean waterbody. Lake Reserv Manage 30: 46–62. [CrossRef] [Google Scholar]
- Moresco GA, Bortolini JC, Dias JD, Pineda A, Jati S, Rodrigues LC. 2017. Drivers of phytoplankton richness and diversity components in Neotropical floodplain lakes, from small to large spatial scales. Hydrobiologia 799: 203–215. [CrossRef] [Google Scholar]
- Nakhate AB, Kale MK. 2018. Studies on physicochemical parameters in Kankaleshwar Lake, Dist. Beed (M.S.) India. Int J Universal Print 4: 347–351. [Google Scholar]
- Nayek S, Gupta S, Pobi KK. 2018. Physicochemical characteristics and trophic state evaluation of post glacial mountain lake using multivariate analysis. Glob J Environ Sci Manag 4: 451–464. [Google Scholar]
- Nejumal KK, Sreejith MV, Dineep D, Aravind UK, Aravindakumar CT. 2021. Identification and ecological hazard analysis of contaminants of emerging concerns (CECs) in water bodies located in a coastal metropolitan environment. Water Air Soil Pollut 232: 1–14. [Google Scholar]
- Nicholls KM, Kennedy W, Hannet C. 1980. Fish-kill in Heart Lake, Ontario, associated with the collapse of a massive population of Ceratium hirundinella. (Dinophyceae). Freshw Biol 10: 553–561. [CrossRef] [Google Scholar]
- Ongun-Sevindik T, Durgut-Kınalı Z, Tunca H. 2023. Temporal and spatial changes in diatom community structure with the effects of environmental parameters, and ecological status assessment by diatom indices in three shallow lakes (Sakarya, Turkey). Biologia 78: 373–387. [Google Scholar]
- Öterler B. 2018. Daily vertical variation in phytoplankton composition of a drinking water reservoir (Kadıköy Reservoir-Edirne) during summer stratification. Trakya Univ J Nat Sci 19: 21–31. [Google Scholar]
- Padisák J. 1985. Population dynamics of the dinoflagellate Ceratium hirundinella in the largest shallow lake of Central Europe, Lake Balaton, Hungary. Freshw Biol 15: 43–52. [Google Scholar]
- Padisák J, Crossetti L, Naselli-Flores L. 2009. Use and misuse in the application of the phytoplankton functional classification: a critical review with updates. Hydrobiologia 621: 1–19. [CrossRef] [Google Scholar]
- Padisák J, Grigorszky I, Borics G, Soroczki-Pinter E. 2006. Use of phytoplankton assemblages for monitoring ecological status of lakes within the Water Framework Directives: the assemblage index. Hydrobiologia 553: 1–14. [CrossRef] [Google Scholar]
- Park JG. 2012. Algal Flora of Korea: Cyanophyta: Cyanophyceae: Chroococcales, Oscillatoriales. Freshwater Cyanoprokaryota II, Volume 5, Number 2. National Institute of Biological Resources Environmental Research Complex, Hwangyeong-ro 42, Seo-gu Incheon, 404–708, Republic of Korea. [Google Scholar]
- Pasztaleniec A, Poniewozik M. 2010. Phytoplankton based assessment of the ecological status of four shallow lakes (Eastern Poland) according to Water Framework Directive − a comparison of approaches. Limnologica 40: 251–259. [CrossRef] [Google Scholar]
- Pednekar SM, Kerkar V, Matondkar SGP. 2014. Spatiotemporal distribution in phytoplankton community with distinct salinity regimes along the Mandovi estuary, Goa, India. Turk J Bot 38: 800–818. [CrossRef] [Google Scholar]
- Pereira LCC, Jiménez JA, Koening ML, Neto FFP, Medeiros C, Da Costa RM, 2005. Effect of coastline properties and wastewater on plankton composition and distribution in a stressed environment on the north coast of Olinda-PE (Brazil). Braz Arch Biol Technol 48: 1013–1026. [CrossRef] [Google Scholar]
- Pérez-Martínez C, Sánchez-Castillo P. 2001. Temporal occurrence of Ceratium hirundinella in Spanish reservoirs. Hydrobiologia 452: 101–107. [CrossRef] [Google Scholar]
- Pérez-Martínez C. and Sánchez-Castillo P. 2002. Winter dominance of Ceratium hirundinella in a southern north-temperate reservoir. J Plank Res 24: 89–96. [CrossRef] [Google Scholar]
- Phillips G, Lyche-Solheim A, Skjelbred B, Mischke U, Drakare S, Free G, Jarvinen M, De Hoyos C, Morabito G, Poikane S. 2013. A phytoplankton trophic index to assess the status of lakes for the Water Framework Directive. Hydrobiologia 704: 75–95. [Google Scholar]
- Poniewozik M, Lenard T. 2022. Phytoplankton Composition and Ecological Status of Lakes with Cyanobacteria Dominance. Int J Environ Res Public Health 19: 3832. [CrossRef] [PubMed] [Google Scholar]
- Ramakrishnaiah CR, Sadashivaiah C, Ranganna G. 2009. Assessment of water quality index for the groundwater in Tumkur Taluk, Karnataka State, India. J Chem 6: 523–530. [Google Scholar]
- Rangel LM, Silva LHS, Arcifa MS, Perticarrari A. 2009. Driving forces of the diel distribution of phytoplankton functional groups in a shallow tropical lake. Braz J Biol 69: 75–85. [CrossRef] [PubMed] [Google Scholar]
- Reynolds CS, Huszar V, Kruk C, Naselli-Flores L, Melo S. 2002. Towards a functional classification of thefreshwater phytoplankton. J Plank Res 24: 417–428. [CrossRef] [Google Scholar]
- Sahu G, Satpathy KK, Mohanty AK, Sarkar SK. 2012. Variations in community structure of phytoplankton in relation to physicochemical properties of coastal waters, southeast coast of India. Indian J Geo-Mar Sci 41: 223–241. [Google Scholar]
- Sako Y, Ishida T, Nishijama T, Hata Y, 1987. Sexual reproduction and cyst formation in the freshwater dinoflagellate Peridinium penardii. Nippon Suisan Gakkaishi 53: 473–478. [CrossRef] [Google Scholar]
- Sanz-Luque E, Chamizo-Ampudia A, Llamas A, Galvan A, Fernandez E. 2015. Understanding nitrate assimilation and its regulation in microalgae. Front Plant Sci 6: 899. [CrossRef] [PubMed] [Google Scholar]
- Sevindik TO, Tunca H, Gönülol A, Gürsoy N, Küçükkaya ŞN, Kınalı Z. 2017. Phytoplankton dynamics and structure, and ecological status estimation by the Q assemblage index: a comparative analysis in two shallow Mediterranean lakes. Turk J Bot 41: 25–36. [CrossRef] [Google Scholar]
- Sharma S, Walia YK. 2017. Water quality investigation by physicochemical parameters of Satluj River (Himachal Pradesh, India). Curr World Environ 12: 174–180. [CrossRef] [Google Scholar]
- Smilauer P. 2012. Canoco5 (version 5.0). Wageningen (Netherlands): Plant Research International. [Google Scholar]
- Søndergaard M, Jeppesen E, Jensen J.P. and Amsinck SL. 2005. Water framework directive: ecological classification of Danish lakes. J Appl Ecol 42: 616–629. [CrossRef] [Google Scholar]
- Sournia A. 1978. Phytoplankton manual. Monographs onoceanographic methodology. UNESCO. [Google Scholar]
- Soylu EN. 2006. An investigation on the phytoplankton of Liman Lake (Samsun-Turkey) and it's seasonal variation. Ondokuzmayıs University, Institute of Science, doctoral thesis no: 16096. 95 p. [Google Scholar]
- SPSS for Windows. 2008. SPSS Statistics Base (version 18.0) for Windows User's Guide, SPSS Inc, Chicago, IL. [Google Scholar]
- Starmach K. 1974. Cryptophyceae-Kryptofity. Dinophyceae-Dinofity. Raphidophyceae-Rafidofity. Państwowe Wydawnictwo Naukowe , ISSN 0071–5840, (1–519p). [Google Scholar]
- Stenger-Kovács C, Buczko K, Hajnal E, Padisák J. 2007. Epiphytic, littoral diatoms as bioindicators of shallow lake trophic status: Trophic Diatom Index for Lakes (TDIL) developed in Hungary. Hydrobiologia 589: 141–154. [Google Scholar]
- Sterrenburg FAS. 1995. Studies on the Genera Gyrosigma and Pleurosigma (Bacillariophyceae); Gyrosigma balticum (Ehrenberg) Rabenhorst, G. pensacolae sp. n. and simulacrum species. Bot Mar 38: 401. [Google Scholar]
- Sun J, Liu D. 2003. Geometric models for calculating cell biovolume and surface area for phytoplankton. J Plankton Res 25: 1331–1346. [CrossRef] [Google Scholar]
- SWQMR. 2021. Regulation on the Revision of the Surface Water Quality Regulation. Official Gazette Number: 31513. Environmental quality standards for some parameters in surface water masses and their usage purposes, Ministry of Agriculture and Forestry, Ankara. [Google Scholar]
- Taşkın E, Akbulut A, Yıldız A, Şahin B, Şen B, Uzunöz C, Solak C, Başdemir D, Sevik F, Sönmez F, Açkgöz I, Pabuccu K, Öztürk M, Alp MT, Albay M, Çakır M, Özbay Ö, Can Ö, Akçaalan R, Atıcı T, et al. 2019. Turkey algae list. Ali Nihat Gökyiğit Foundation Publication, İstanbul. 804 pp. [Google Scholar]
- Ter Braak CJF, Šmilauer P. 2012. Canoco reference manual and user's guide: software for ordination, version 5.15. Microcomputer Power, Ithaca, USA, 496 p. [Google Scholar]
- Török P, T-Krasznai E, B-Béres V, Bácsi I, Borics G,Tóthmérész B. 2016. Functional diversity supports the biomass-diversity humpedback relationship in phytoplankton assemblages. Funct Ecol 30: 1593–1602. [CrossRef] [Google Scholar]
- Utermöhl H. 1958. Zur Ver vollkommnung der quantitativen Phytoplankton-Methodik. Mitteilungen der Internationale Vereinigung der theoretische und Angewandte Limnologie 5: 567–596. [Google Scholar]
- Vadrucci MR, Barbone E, Ungaro N, Romano A, Bucci R. 2017. Application of taxonomic and morpho-functional properties of phytoplankton communities to water quality assessment for artificial lakes in the Mediterranean Ecoregion. J Plank Res 39: 550–563. [CrossRef] [Google Scholar]
- Vidaković D, Krizmanić J, Ndoj E, Miho A, Kupe L, Schneider SC. 2020. Changes in the diatom community in the great lake (Lurë National Park, Albania) from 2005 to 2017 and first steps towards assessment the water quality. Biologia 75: 1815–1824. [CrossRef] [Google Scholar]
- Viet ND, Bac NA, Huong HTT. 2016. Dissolved oxygen as an ındicator for eutrophication in freshwater lakes. Proc Int Conference on Environmental Engineering and Management for Sustainable Development. Hanoi, Vietnam. [Google Scholar]
- Walker WW. 1982. An empirical analysis of phosphorus, nitrogen, and turbidity effects on reservoir chlorophyll-a levels. Can Water Resour J 7: 88–107. [CrossRef] [Google Scholar]
- Watanabe M, Watanabe M, Fukuyo Y, 1982. Encysment and excystment of red tide flagellates I. Introduction of encystment of Scrippsiella trochoidea. Res Rep Nat Inst Environ Stud 30: 27–42. [Google Scholar]
- Wu T, Wang S, Su B, Wu H, Wang G. 2021. Understanding the water quality change of the Yilong Lake based on comprehensive assessment methods. Ecol Indic 126: 1–9. [Google Scholar]
- Xiao L, Wang T, Hu R, Han B, Wang S, Qian X, Padisák J. 2011. Succession of phytoplankton functional groups regulated by monsoonal hydrology in a large canyon-shaped reservoir. Water Res 45: 5099–5109. [CrossRef] [PubMed] [Google Scholar]
- Yang M, Bi Y, Hu J, Zhu K, Zhou G, Hu Z. 2011. Seasonal variation in functional phytoplankton groups in Xiangxi Bay, Three Gorges Reservoir. Chin J Oceanol Limnol 29: 1057–1064. [CrossRef] [Google Scholar]
- Zanco BF, Pineda A, Bortolini JC, Jati S, Rodrigues LC. 2017. Phytoplankton functionalgroups indicators of environmental conditionsin floodplain rivers and lakes of the Paraná Basin. Acta Limnolog Bras 29: e119. [Google Scholar]
- Zelinka M, Marvan P. 1961. Zur Präzisirung der biologischen Klassifikation der Reinheit fliessender Gewässer. Arch Hydrobiol 57: 389–407. [Google Scholar]
Cite this article as:Maraşlıoğlu F, Coşkun T, Çetin T, Kağnıcıoğlu N, Ekmekçi F, Şahin M. 2023. Use of different indices to assess the ecological status of lake systems in the eastern mediterranean river basin. Int. J. Lim. 59: 9
All Tables
Spearman rank correlation between indices and environmental parameters (N = 87). The meanings of the coefficients in the table is as follows: very weak or no association (±0.0 to ±0.2), weak association (±0.2 to ±0.4), moderate association (±0.4 to ±0.6), strong association (±0.6 to ±0.8), very strong association (±0.8 to ±1.0).
Ecological status of the seven sites (lakes) according to various biotic and abiotic indices. The meaning of different colors used in the table is as follows: blue − high quality, green − good quality, yellow − moderate quality, orange − poor quality, red − bad quality.
All Figures
 |
Fig. 1 Location map of the study area in the Eastern Mediterranean River basin. |
| In the text | |
 |
Fig. 2 PCA diagram of sampling sites (circle) and environmental parameters (arrow) relationships. Water temperature (Temp), electrical conductivity (EC), suspended solids (SS), ammonium (NH4+), nitrite (NO2), nitrate (NO3), total nitrogen (TN), sulfate (SO4), total phosphorus (TP), dissolved oxygen (DO), biological oxygen demand (BOD5), chemical oxygen demand (COD), total organic carbon (TOC), and potential hydrogen (pH). The full names and abbreviation codes of the lakes are given in Table 1. |
| In the text | |
 |
Fig. 3 CCA diagram of species (circle) and environmental parameters (arrow) relationships. Water temperature (Temp), electrical conductivity (EC), suspended solids (SS), ammonium (NH4+), nitrite (NO2), nitrate (NO3), total nitrogen (TN), sulfate (SO4), total phosphorus (TP), dissolved oxygen (DO), biological oxygen demand (BOD5), chemical oxygen demand (COD), total organic carbon (TOC), and potential of hydrogen (pH). The full names abbreviation codes of 32 taxa are given in Table 2. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


