| Issue |
Int. J. Lim.
Volume 62, 2026
|
|
|---|---|---|
| Article Number | 1 | |
| Number of page(s) | 11 | |
| DOI | https://doi.org/10.1051/limn/2025012 | |
| Published online | 11 February 2026 | |
Research article
Characterisation of the nuptial colouration pattern of Phoxinus lumaireul Schinz, 1840 (Teleostei, Leuciscidae) and first record in France
1
Unité Patrimoine Naturel – Centre d’expertise et de données (UAR 2006 OFB– MNHN – CNRS), Muséum national d’Histoire naturelle, 36 rue Geoffroy-Saint-Hilaire CP 41, 75005 Paris, France
2
Office Français de la Biodiversité, Direction Régionale Auvergne-Rhône-Alpes, Unité Spécialisée Milieux Lacustres, Pisciculture de Rives, 13 Quai Georges Pianta, 74200 Thonon-les-Bains, France
3
Museo Civico di Storia Naturale, Laboratorio di Ittiologia, Via S. Francesco di Sales 188, 10022 Carmagnola TO, Italy
* Corresponding author: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
4
July
2025
Accepted:
11
December
2024
Abstract
Recent molecular studies have highlighted the taxonomical complexity of the Eurasian minnows Phoxinus spp, pointing out their multiple endemic species to their respective catchments, and also the multiple translocations between drainages. Here, we report the first record of the Italian minnow Phoxinus lumaireul in France: a male specimen was caught in the Lake Geneva at Publier (Haute-Savoie department) the 31st May 2010 during an elecrofishing survey monitored by the ONEMA. The identification was done from a picture, according to morphological observations and the nuptial colouration pattern, in comparison with P. lumaireul from the Po catchment. We then attest the presence of a third minnow species in the Lake Geneva basin, in addition to P. csikii and P. septimaniae. The nuptial colouration pattern of P. lumaireul is diagnosable by very little developed nuptial tubercles on the head, pectoral fins yellowish hyaline, a green bluish stripe on Z3 until the caudal peduncle origin and black beyond, Z4 green yellowish and a belly shiny red from the throat to the caudal fin base for males, and pectoral, pelvic and anal fin bases pinkish, yellow spot on the operculum, Z4 slightly golden and Z5 white for female. Our morphological identification corroborates a molecular study from the Swiss part of the Lake Geneva basin. Phoxinus lumaireul is then the seventh and the first non-native minnow species occurring in France. Its introduction through human translocations seems to be ancient. We also discussed about ecological impacts for French and Swiss managers.
Key words: Cryptic invasion / Italian minnow / Lake Geneva / Morphology / Nuptial colouration
© G.P.J. Denys et al., published by EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
1 Introduction
Eurasian minnows Phoxinus spp are small, gregarious, rheophilic and cryophilic leuciscids inhabitants of streams and clear lakes from Ireland and the Iberian Peninsula to Korea (Bănărescu & Coad, 1991; Esposito et al., 2024a). They are generally described as having an olive-green colouration, a lighter longitudinal line above dark blotches on the flanks and a white-grey belly (e.g., Kottelat, 2007). But during the reproductive season, nuptial tubercles on the head and throat appear (Witkowski & Rogowska, 1992; Chen & Arratia, 1996), as well as nuptial colourations with shiny colours on the body (e.g., Denys et al., 2020).
The taxonomic knowledge about this genus has considerably changed during the last two decades, with numerous species descriptions and revalidations of species names (see Esposito et al., 2024a). However, the species delineations are also supported by molecular data and also morphological characters such as the snout and anal fin shapes, the presence of scales between the pectoral fins, the number of scales in the lateral series as well as different morphometrics data (Kottelat, 2007; Palandačić et al., 2017, 2024a; Bogutskaya et al., 2020, 2023; Denys et al., 2020; Turan et al., 2023; Artaev et al., 2024a,b, 2025; Bayçelebi et al., 2024; Bayçelebi, 2025).
However, not only these morphological characters are difficult (even impossible) to observe in the field on alive fish. But also, minnow species present some phenotypic plasticities (Bianco & De Bonis, 2015; Collin & Fumagalli, 2011, 2015; Palandačić et al., 2017; Ramler et al., 2017; Bogutskaya et al., 2020; Scharnweber, 2020). All this means that riverine managers have the feeling of not having reliable diagnostic criteria to identify the different species of minnows, with as consequence the absence of the minnow species diversity into their management plans. Denys et al. (2020) then proposed an alternative to morphological criteria: the use of nuptial colouration patterns. They characterised the nuptial colouration pattern of both male and female of each evolutionary lineage in France from molecular data, allowing the delineation of six minnow species: Phoxinus phoxinus (Linnaeus, 1758) sensu stricto in the Rhine, Meuse and Seine drainages, Phoxinus bigerri Kottelat, 2007 in the Adour drainage, Phoxinus septimaniae Kottelat, 2007 in the Mediterranean basin, Phoxinus csikii Hankó, 1922 in the Rhine drainage, as well as Phoxinus fayollarum Denys, Dettai, Persat, Daszkiewicz, Hautecœur & Keith, 2020 and Phoxinus dragarum Denys, Dettai, Persat, Daszkiewicz, Hautecœur & Keith, 2020 which are respectively endemic to the Loire and the Garonne drainages. Further taxonomic revisions on other minnow species also demonstrated other nuptial colouration patterns which could be diagnostic (Bogutskaya et al., 2023; Turan et al., 2023; Artaev et al., 2024a; Bayçelebi, 2025).
In the Lake Geneva basin, located in the Rhône River at the French-Swiss border, minnows are considered as rare (Jurine, 1825; Raymond, pers. com.). Nevertheless, the presence of two Phoxinus species is attested: P. csikii and P. septimaniae (Palandačić et al., 2017; Denys et al., 2020). Alexander & Seehausen (2021), during their DNA Barcoding of the whole of fish species in Swiss lakes program (Projet Lac), also detected a third minnow species in the Stockalper Canal using molecular data: the Italian minnow P. lumaireul (Schinz, 1840). This species is endemic to the Adriatic basin, including the Po River – its type locality (Palandačić et al., 2017, 2020). In May 2010, the Office National de l’Eau et des Milieux Aquatiques (ONEMA; now called French Office of Biodiversity, OFB) conducted an electrofishing campaign in the Lake Geneva, catching a minnow on the 31st May 2010. Because of its rarity and its shiny breeding colourations, the fish was photographed before release. Several years later, the picture was sent to the Muséum national d’Histoire naturelle (MNHN, Paris) for identification.
The aim of this study is to identify this minnow with morphological criteria but also according to its nuptial colouration pattern. In order to do this, we will characterise the nuptial colouration pattern of P. lumaireul and we will compare it to those of P. csikii and P. septimaniae (Denys et al., 2020). This study will then report the first record of P. lumaireul in France, being then the seventh minnow species and the first allochtonous one. We will also discuss about the consequence of its introduction on native minnow species and on the management.
2 Material and methods
2.1 Sampling
The French part of the Lake Geneva has been monitored for the first time by the ONEMA in 2010. The location at Publier (46°23’46” N, 6°32’22” E) is one of the three sites surveyed (Raymond et al., 2010). The inventory consisted to catch fishes using multi-mesh gill nets according to the protocol DCE EN14757, as well as a prospecting of the riparian zone with depth inferior of 2 meters, by electrofishing following the punctual abundance sampling method, after mapping the aquatic habitats beforehand allowing the number of sampling points to be distributed per habitat (Degiorigi et al., 2001). The water temperature was 14.8°C, whereas the substrate was composed in decreasing order by blocks, sand-silt, pebbles, gravels, afference zones, scattered helophytes and woody supports. Fish caught were conserved in a white bucket in order to limit the water warming by the sun, before being measured, photographed and released.
Concerning the Italian minnows from the Po catchment, fish were caught by electrofishing or hand net from June 2011 to June 2016 from four locations: Varaita Torrent at Costigliole Saluzzo (44°33’49’’N – 7°28’29’’E), Sangone Torrent at Trana (45°01’43’’N – 7°23’42’’E), Chisone Torrent at Pinerolo (45°51’42’’N – 7°20’41’’E) and Lago Laus at Roreto Chisone (45°03’00’’N – 7°08’08’’E). Alive and euthanised fish were then photographed after capture. Fish samples were genotyped in previous studies attesting their affiliation to P. lumaireul (Geiger et al., 2014; Ramler et al., 2017; Palandačić et al., 2017; De Santis et al., 2021) (Tab. 1; Fig. 1). Twelve photos including 27 specimens (22 males and 7 females) composed our material to characterise the nuptial colouration pattern of P. lumaireul (Fig. 2; Supplementary data 1).
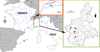 |
Fig. 1 Sampling locations (red squares) of minnows in the Po catchment in the Italian Piedmont (green box) where specimens of P. lumaireul were caught and used for the nuptial colouration characterisation (see Tab. 1, Figs. 2, 4b, and 5c), as well as in the French part of the Lake Geneva (red box) where the minnow specimen of unknown species was photographed and presented in Fig. 4a. Locations: 1) Varaita Torrent at Costigliole Saluzzo 44°33’49’’N – 7°28’29’’E, 2) Chisone Torrent at Pinerolo 45°51’42’’N – 7°20’41’’E, 3) Lago Laus, Roreto Chisone 45°03’00’’N – 7°08’08’’E, 4) Sangone Torrent at Trana 45°01’43’’N – 7°23’42’’E, 5) Lake Geneva at Publier 46°23’46”N – 6°32’22”E. |
 |
Fig. 2 Phoxinus lumaireul in the Po catchment with alive nuptial colourations: male (68 mm TL) from the Varaita Torrent at Costigliole Saluzzo (a), five males (57 to 64 mm TL) from the Chisone Torrent at Pinerolo (b), male (not measured) from the Lago del Lau at Roreto Chisone (c), two females (75 mm TL (d) and 81 mm TL (e)) from the Sangone Torrent at Trana, nuptial colouration pattern for male (f) and female (g); photo credits: G. Delmastro. |
Origin of Italian minnows genotyped as P. lumaireul according to previous studies, and used for the characterisation of the nuptial colouration pattern. Museums abbrevations: Museo Civico di Storia Naturale di Carmagnola, Carmagnola, Italy (MCCI); Muséum national d’Histoire naturelle, Paris, France (MNHN); Naturhistorisches Museum, Zoologische Abteilung, Fische, Vienna, Austria (NMW); Zoologisches Forschungsmuseum Alexander König, Bonn, Germany (ZFMK).
2.2 Sexual determination
The sex of photographed specimens was identified on the pictures using external anatomy i.e. length and shape of the pectoral fin. A male minnow has a long, wide, thick and rounded pectoral fin, whereas the female’s one is short and less rounded (Frost, 1943). Moreover, during the breeding period, female specimens are also identifiable by their gravid belly. These characteristics are only applicable for adult fish.
2.3 Morphological identification
The three species are P. csikii, P. lumaireul and P. septimaniae discriminated by about 15 morphological characters (Kottelat and Freyhof, 2007; Palandačić et al., 2017; Denys et al., 2020) (see Supplementary data 2). However, only five characters (the snout shape, the snout length (% head length HL), the position of the mouth, the anal fin margin and the ratio length/depth of the caudal peduncle) were visible and used for the morphological identification. Approximate lengths ratios (snout length and caudal peduncule depth) were done using pixels numbers counted on the photographs of the single Lake Geneva specimen (Supplementary data 3) using the GIMP software (https://www.gimp.org/).
2.4 Characterisation of the nuptial colouration pattern of P. lumaireul
We characterised separately the nuptial colouration pattern of both males and females P. lumaireul observing living animals in aquariums and also after electrofishing and handling (Tab. 1). We follow the terminology of Denys et al. (2020) (Fig. 3a) with five coloured zones Z1 to Z5 from the back to the belly, for the description of the nuptial coat for male and female. The observed and characterized patterns were after compared to those of P. csikii and P. septimaniae for which the discriminations for both males and females between the two species are truly reliable (Denys et al., 2020) (Fig. 3b,c).
 |
Fig. 3 The five zones on the minnow’s flank characterising the nuptial colouration pattern (a) following Denys et al. (2020), and both male and female nuptial colouration patterns for respectively Phoxinus csikii (b) and Phoxinus septimaniae (c) according to Denys et al. (2020); drawings adapted and modified from Denys et al. (2020). |
 |
Fig. 4 Male minnow (65 mm TL, a) in nuptial colouration with an unknown species origin caught in the Lake Geneva at Publier the 31st May 2010 and one male Phoxinus lumaireul (83 mm TL, b; identified by Geiger et al. (2014)) from the Sangone Torrent at Trana the 15th June 2011; photo credits: respectively J.-C. Raymond / OFB and G. Delmastro. |
3 Results and discussion
3.1 Nuptial colouration pattern of Phoxinus lumaireul
Phoxinus lumaireul is characterised by the following nuptial colouration pattern (Fig. 2):
Male (Fig. 2a,b,c,f): Lips and pectoral, pelvic and anal fin bases red, these fins yellowish hyaline. Nuptial tubercles on the head very weakly expressed. Red dot on the dorsal fin base. Snout and top of head dark greyish with red dots above the eyes. Cheeks white greyish. Operculum with a white spot at the uppermost corner followed by a yellow spot on the supraoperculum. Z1 and Z2 brownish overlayed with blackish bars. Green blueish stripe until the caudal-peduncle origin and black beyond along Z3. Z4 green yellowish. Z5 black between pectoral fin and anal fin base. Belly shiny red from the throat to the caudal fin base.
Female (Fig. 2d,e,g): Nuptial tubercles on the head quasi absent. Less brightly coloured, pectoral, pelvic and anal fin bases pinkish, these fins yellow. Z1 and Z2 brownish with black vermiculations, rather than stripes. Black stripe along Z3. Z4 slightly golden. Z5 white or silvery. Belly white.
Phoxinus lumaireul is native to the North Adriatic Sea basin, from the Esino and Potenza catchments (central Apennines) to the Western Balkans (Kottelat & Freyhof, 2007; Palandačić et al., 2015, 2017; Vucić et al., 2018; Caputo Barucchi et al., 2022). Molecular data highlight at least six evolutionary lineages within this species (Palandačić et al., 2015, 2017; Vucić et al., 2018, 2022). Moreover, some morphological variabilities were observed among populations living in Western Balkans (Bianco & De Bonis, 2015; Palandačić et al., 2017; Bogutskaya et al., 2020). Our samples came from the Po catchment, the type locality, and are affiliated to the clade 1a sensu Palandačić et al. (2017). Thus, our diagnosis might be applicable only for this lineage, as we have no data about the nuptial colouration pattern from populations affiliated to the other lineages.
Comparing it to the two other minnow species of the perialpine lakes, Phoxinus lumaireul is then distinguishable from P. csikii by very little developed nuptial tubercles on the head (vs. well developed), a green bluish stripe on Z3 until the caudal peduncle origin (vs. green or blackish stripe fractioned in blotches) and a Z4 green yellowish (vs. green) for males (Fig. 3b), and a Z4 slightly golden (vs. golden) for females (Fig. 3b) (Denys et al., 2020). It is also distinguishable from P. septimaniae by very little developed nuptial tubercles on the head (vs. well developed), the absence of a yellow spot between the eye and the mouth (vs. presence of a yellow spot), pectoral fins yellowish hyaline (vs. yellowish-orange with a red disc at inners side), a green bluish stripe on Z3 until the caudal peduncle origin and black beyond (vs. blackish stripe until the caudal fin origin), Z4 green yellowish (vs. greyish, without green colouration, except rarely around pectoral fin), and a belly shiny red from the throat to the caudal fin base (vs. white) for males (Fig. 3c), pectoral, pelvic and anal fin bases pinkish (vs. no pigmentation), presence of a yellow spot on the operculum (vs. no or an indistinct yellow spot on the operculum), Z4 slightly golden (vs. silvery white) and Z5 white (vs. black) for females (Fig. 3c) (Denys et al., 2020).
Our observations in aquariums allowed the characterisation of the colouration patterns of the dorsal fin with a red spot on its basis. Denys et al. (2020) described nuptial colourations patterns on euthanised fish fitted on a shelf, which did not allow to see well the dorsal fin. So, this last character was not used to describe the nuptial colouration patterns of the six French minnow species. An observation in aquariums would be helpful to complete gap as well as to find other characters, and better resolve the taxonomy of this genus (Denys et al., 2025).
3.2 The minnow from the Lake Geneva
One male minnow (65 mm total length (TL); Fig. 4a) was caught at Publier (Haute-Savoie department, France) along seven other fish species (Perca fluviatilis Linnaeus, 1758, Rutilus rutilus (Linnaeus, 1758), Salariopis fluviatilis (Asso, 1801), Gobio gobio (Linnaeux, 1758), Salmo trutta Linnaeus, 1758, Cottus gobio Linnaeus, 1758, Squalius cephalus (Linnaeus, 1758)) and the signal crayfish Pacifastacus leniusculus (Dana, 1852) (Raymond et al., 2010). It was caught in bank rip-rap at 0.9 m of depth, not far from a tributary of Lake Geneva.
We attempted to identify the sample using morphological characters on photos provided by the ONEMA (Fig. 4a, Supplementary data 3). The following morphological data allows to identify the minnow from the Lake Geneva as P. lumaireul: combination of the very stout snout shape, the snout length 26% HL and the ratio length/depth of the caudal peduncle equal to 2.48 (Tab. 2). It therefore differs from P. septimaniae by its very stout snout shape and a shorter snout length. Similarly, it differs from P. csikii by a slightly smaller ratio length/depth of the caudal peduncle. Nevertheless, the different way of measurement (pixel vs. calliper) can induce a bias in the results (e.g., Takács et al., 2016) and then a misinterpretation risk. However, this approach was already helpful for taxonomic expertise on photos (e.g., Cuinet et al., 2024). Finally, our results are congruent with those of Ramler et al. (2017) who already highlighted that Italian minnows (namely P. lumaireul) could be discriminated from the Danubian populations (namely P. csikii) by the caudal peduncule depth.
The colouration pattern of the minnow photographed in Lake Geneva (Fig. 4a) is less shiny than those observed in other pictures of P. lumaireul (Fig. 2). Nonetheless its general nuptial colouration pattern is consistent with that of P. lumaireul as it was detailed above and in Figure 2. The remarkable character is the shiny red belly which is persistant, whereas colours in Z1-Z5 are generally less strongly expressed. The fish was caught by electrofishing and handled. These events are sources of stress wich disrupts the neuro-hormonal functioning of the skin pigmentation (e.g., Bernier et al., 2009; Vissio et al., 2021). Our minnow was indeed conserved in a white bucket, inducing then melanosomes aggregation due to a diminution of the cells size within minnows (Abbott, 1973; Amiri, 1991). This new white environment induced a mechanism of background adaptation, thus affecting the melanogenesis (see Fujii, 2000; Leclercq et al., 2010), hence the absence of shiny colour pattern. However, the red belly is still well marked and seems to still be diagnostic. This colouration is similar to a P. lumaireul minnow caught in the Sangone Torrent at Trana (Fig. 4b).
Morphological observations and approximated lengths ratios taken on photograph (Fig. 4; in pixel numbers) of the male minnow caught in the Lake Geneva the 31st May 2010 compared to data from Kottelat and Freyhof (2007) (*), Palandačić et al. (2017) ($) and Denys et al. (2020) (£) on P. csikii, P. lumaireul and P. septimaniae from morphomeristic analyses with measurements issued from digital calliper; diagnostic characters are in bold; for the snout shape see the Supplementary data 2; HL means head length.
3.3 First photographic evidence of P. lumaireul in France
The morphological observation and the nuptial colouration pattern corroborate the identification of the minnow caught in the Lake Geneva the 31st May 2010 as Phoxinus lumaireul. According to morphological data, this study confirms the molecular detection in the Stockalper Canal, a Swiss tributary of the Lake Geneva, and the presence of three minnow species in the Lake Geneva basin with P. csikii and P. septimaniae, as in other Swiss and Italian perialpine lakes (Alexander & Seehausen, 2021; De Santis et al., 2021). However, minnows should be considered as occurring in the tributaries of Lake Geneva, as they are only rare in the Lake (Jurine, 1825). Nevertheless, hybridisation with P. csikii and P. septimaniae is known (Palandačić et al., 2017; De Santis et al., 2021). So, it might be possible that this specimen could be in fact a hybrid, but that it is unlikely as the recorded morphological characteristics clearly match P. lumaireul from other populations.
Forel (1904) hypothetised that minnows, like most other fish species, were introduced in the Lake Geneva. However, considering the biogeographical context of the Lake Geneva, with the current and ancient connections with basin shifts between the Rhône and the Rhine catchments since the last Ice age (see Persat et al., 2020), P. csikii and P. septimaniae seem to be native. At our knowledges, the only connection between the Rhône and the Po catchment occurred though the Durance river (southeastern France) during the late Pleistocene (Splendiani et al., 2020). So the presence of P. lumaireul in the Lake Geneva must have an anthropogenic origin. Two other Italian fish species introduced in the Lake Geneva are known: the southern pike Esox cisalpinus Bianco & Delmastro, 2011 and the southern rudd Scardinius hesperidicus Bonaparte, 1845 (Denys et al., 2014; Keith et al., 2020; Alexander & Seehausen, 2021). All three species are native from the Padano Veneto region including the Po drainage (Lorenzoni et al., 2019). Thus, P. lumaireul is then the seventh one, and the first allochtonous minnow species occurring in France, whereas this species is native in Switzerland but only in the Po drainage where minnows occure in few localities according to Pedroli et al. (1991). This species will then be listed in the French taxonomic register TAXREF (TAXREF, 2025) of the National Inventory of Natural Heritage (Inventaire du Patrimoine Naturel – INPN).
Minnows introductions are essentially due to their use as live bait for the angling of brown trout Salmo trutta Linnaeus, 1758 (Banha et al., 2016), as it was already documented through Europe (Museth et al., 2007; Miró & Ventura, 2015; Palandačić et al., 2015, 2017, 2020, 2022; Corral-Lou et al., 2019; Denys et al., 2020; Garcia-Raventós et al., 2020; Alexander & Seehausen, 2021; De Santis et al., 2021; Esposito et al., 2024b; Sternberg et al., 2025), as well in Switzerland (Pedroli et al., 1991). So, a recent introduction is plausible. However, an ancient introduction cannot be excluded. Palandačić et al. (2020) already highlighted minnow translocations during the 19th century. According to Denys et al. (2014), the introduction of the southern pike in the Lake Geneva might be due to a trade between the Geneva monastery and the Aosta church during the 12th century. Lunel & Lunel (1874) described several nuptial colouration patterns of minnows from the Lake Geneva basin. Denys et al. (2020) identified one as P. csikii (Fig. 5a). However, the drawning presented in the Figure 5b seems to have the nuptial colouration pattern of P. lumaireul and looks like a male minnow from the Chisone Torrent in the Po catchment (Fig. 5c). So, the introduction of P. lumaireul in the Lake Geneva would be ancient.
 |
Fig. 5 Minnows drawings (a,b; modified symmetrically to the left side) from Lunel and Lunel (1874): a male identified as Phoxinus csikii by Denys et al. (2020) (a), another male having the nuptial coulouration pattern of Phoxinus lumaireul (b) like the male (57 mm TL) from the Chisone Torrent at Pinerolo (c; photo credits: G. Delmastro). |
3.4 Consequence for the management of the Lake Geneva
Introduction of a new allochtonous fish species in any environment always induces threats for native fauna such as competition, hybridisation and diseases transmissions (e.g., Gozlan et al., 2010; Cucherousset & Olden, 2011; Ribeiro & Leunda, 2012). Allochtonous minnow’s introduction in one hand have never had a positive impact on brown trout population (Tiberti et al., 2022), and in another hand, have had a negative impact on native species (Museth et al., 2007; Miró et al., 2018; Osorio et al., 2022), as well as on native minnows’ species. Some of these threats are already documented such as hybridisation (Palandačić et al., 2017, 2020, 2024a; Corral-Lou et al., 2019; De Santis et al., 2021, Vucić et al., 2022) and pathogen transmissions (e.g., Esposito et al., 2024a,b). Moreover, the mechanisms of establishement and invasiveness of an allochtonous minnow species is driven by a large trophic niche, a wide physiological tolerance, better immune response as well as a resistance to parasites and a genetic diversity (Thaulow et al., 2013; Scharnweber, 2020; Cruz et al., 2022). Even if the parasitofauna of P. lumaireul has been very little studied (see Esposito et al., 2024a) and no knowledges are known about the trophism, it has therefore a phenotypic plasticity allowing it to adapt to its environment (Bianco & De Bonis, 2015; Ramler et al., 2017; Palandačić et al., 2017), and above all a high genetic variability (Geiger et al., 2014; Palandačić et al., 2015, 2017, 2020; Vucić et al., 2018; De Santis et al., 2021; Caputo Barucchi et al., 2022; Reier et al., 2022, 2025). So, some studies already pointed out its ability to migrate and to colonise new environments including karstic caves (Palandačić et al., 2015, 2020; Reier et al., 2022, 2025), which suggests that P. lumaireul appears to be a good candidate to be an invader minnow species. Fortunately, minnows are rare on the Lake Geneva coast, which requires some prospecting effort using for example environmental DNA (Blackman et al., 2022) or citizen sciences (Palandačić et al., 2024b) in order to get occurrences and knowledges about the three minnows’species from the Lake Geneva.
4 Conclusion
As time passes and the taxonomic knowledge increases, minnows start to become a baffling problem for riverine managers. They previously managed only had to consider a single species in their management efforts, but are now facing the problem of having to manage more species, which are often restricted and endemic to only a few drainages (see Esposito et al., 2024a). Moreover, past introductions mean that today they have to consider allochtonous and potentially invasive species, and then with cryptic characters making them difficult to distinguish. However, our results confirms the observation of Denys et al. (2020) on French populations, that nuptial colouration patterns are diagnostic to distinguish minnow species and a precious character to allow managers to identify the minnows in the field during the breeding period.
We also confirm the presence of three minnow species in the Lake Geneva basin. The list of freshwater fish species occurring in this lake must be updated as well as both French and Swiss legislation. Further investigations are needed to understand the interactions between the three minnow species occurring in the lake, as well as the potential risks on the native fauna.
Acknowledgments
This work was supported by the MNHN, the UAR Patrinat 2006 and the French Office of Biodiversity (OFB). We warmly thank M. Baldeck, L. Giusti, F. Renaudon (Specialised Lake Environment Unit; ONEMA) as well as S. Parussatti (Regional delegation Rhône-Alpes; ONEMA) for their participation to the sampling operation in the Lake Geneva. Italian populations of minnow were collected by electrofishing under scientific permits issued by the Ufficio Caccia Pesca Parchi della Provincia di Cuneo and Tutela Flora e Fauna della Città metropolitana di Torino. Finally, we warmly thank M. Hautecœur for checking the English.
Supplementary Material
Supplementary data 1. Photos used to characterize the nuptial colouration pattern of Phoxinus lumaireul: Varaita Torrent at Costigliole Saluzzo (photo 1 – 5), Sangone Torrent at Trana (6 – 7), Chisone Torrent at Pinerolo (9 – 11) and Lago Laus at Roreto Chisone (12 – 13); photo credits: G.B. Delmastro.
Supplementary data 2. Characters used in the literature for distinguishing P. csikii, P. lumaireul and P. septimaniae according to Kottelat and Freyhof (2007), Palandačić et al. (2017), Denys et al. (2020) and this study; credits photos: Denys & Delmatro.
Supplementary data 3. Pictures of the male minnow caught in the Geneva Lake at Publier the 31st May 2010, focusing on the head and the caudal peduncle on which the head length (HL), the snout length (SnL) as well as the caudal peduncle length (CPL) and depth (CPD) were measured (grey lines); photo credits: J.-C. Raymond / OFB.
Access Supplementary MaterialReferences
- Abbott FS. 1973. Endocrine regulation of pigmentation in fish. Amer Zool 13: 885–894. [Google Scholar]
- Alexander T, Seehausen O. 2021. Diversity, Distribution and Community Composition of Fish in Perialpine Lakes. “Projet Lac” synthesis report. Eawag: Swiss Federal Institute of Aquatic Science and Technology, 282 p. [Google Scholar]
- Amiri M. 1991. The effects of spinal nerve section on responses of melanophores in the minnow, Phoxinus phoxinus (L.). J Fish Biol 39: 71–82. [Google Scholar]
- Artaev O, Turbanov I, Bolotovskiy A, Gandlin A, Levin B. 2024a. Taxonomic revision of Phoxinus minnows (Leuciscidae) from Caucasus, with description of a new narrow-ranged endemic species. Zoosyst Evol 100: 291–308. [Google Scholar]
- Artaev O, Bolotovskiy A, Turbanov I, Gandlin A, Kutuzov A, Levina M, Melentev D, Pozdeev I, Borisov M, Levin B. 2024b. Forgotten for two centuries: redescription of Phoxinus isetensis (Georgi, 1775) (Cypriniformes, Leuciscidae) – the most widespread minnow in Europe. Zoosyst Evol 100: 1155–1173. [Google Scholar]
- Artaev O, Turbanov I, Bolotovskiy A, Gandlin A, Levin B. 2025. A new super narrow-ranged endemic Phoxinus minnow (Leuciscidae) from the Caucasus. Zoosyst Evol 101: 1137–1154. [Google Scholar]
- Bănărescu P, Coad BW. 1991. Cyprinids of Eurasia. In: Winfield IJ. and Nelson JS. (eds.), Cyprinid Fishes Systematics, Biology and Exploitation. Fish and Fisheries, series 3, London, Now York, Tokyo, Melbourne, Madras: Chapman & Hall, pp. 127–155. [Google Scholar]
- Banha F, Diniz AM, Anastácio P. 2016. The role of angler’s perceptions and habits in biological invasions: perspectives from Iberian Peninsula. Aquat Conser Mar Freshw Ecosyst 27: 51–64. [Google Scholar]
- Bayçelebi E. 2025. Discovery of a new minnow species in the Black and Marmara Sea basins, Türkiye. Zoosyst Evol 101: 1155–1162. [Google Scholar]
- Bayçelebi E, Aksu İ, Turan D. 2024. Description of a new species of Phoxinus from the Ergene River (Aegean Sea Basin) in Türkiye (Actinopterygii, Leuciscidae). Zoosyst Evol 100: 101–110. [CrossRef] [Google Scholar]
- Bernier N, Flik G, Klaren P. 2009. Regulation and contribution of the corticotropic, melanotropic and thyrotropic axes to the stress response in fishes. In: Bernier NJ, Van Der Kraak G, Farrell AP, Brauner CJ, (eds.), Fish Neuroendocrinology, Amsterdam, Boston: Elsevier/Academic Press, pp 235–311. [Google Scholar]
- Bianco PG, De Bonis S. 2015. A taxonomic study on the genus Phoxinus (Actinopterygii, Cyprinidae) from Italy and western Balkans with description of four new species: P. ketmaieri, P. karsticus, P. apollonicus and P. likai. Res Wildl Conserv 4: 1–17. [Google Scholar]
- Blackman R, Walser J, Rüber L, Brantschen J, Villalba S, Brodersen J, Seehausen O, Altermatt F. 2022. General principles for assignments of communities from eDNA: Open versus closed taxonomic databases. Environ DNA 5: 326–342. [Google Scholar]
- Bogutskaya NG, Jelić D, Vucić M, Jelić M, Diripasko OA, Stefanov T, Klobučar G. 2020. Description of a new species of Phoxinus from the upper Krka River (Adriatic Basin) in Croatia (Actinopterygii: Leuciscidae); first discrovered as a molecular clade. J Fish Biol 96: 378–393. [CrossRef] [PubMed] [Google Scholar]
- Bogutskaya NG, Diripasko OA, Palandačić A. 2023. Novel data support validity of Phoxinus chrysoprasius (Pallas, 1814) (Actinopterygii, Leuciscidae). Eur J Taxon 861: 1–20. [CrossRef] [Google Scholar]
- Caputo Barucchi V, Marconi M, Splendiani A, Casari S, Girardi M, Gandolfi A. 2022. Mitochondrial DNA suggests uniqueness of an isolated population of the Italian minnow (Phoxinus lumaireul Schinz, 1840) (Teleostei: Cyprinidae) in central Apennines (Italy). Eur Zool J 89: 711–718. [Google Scholar]
- Chen XY, Arratia G. 1996. Breeding tubercles of Phoxinus (Teleostei: Cyprinidae): morphology, distribution, and phylogenetic implications. J Morphol 228: 127–144. [Google Scholar]
- Collin H, Fumagalli L. 2011. Evidence for morphological and adaptive genetic divergence between lake and stream habitats in European minnows (Phoxinus phoxinus, Cyprinidae). Mol Ecol 20: 4490–4502. [Google Scholar]
- Collin H, Fumagalli L. 2015. The role of geography and ecology in shaping repeated patterns of morphological and genetic differentiation between European minnows (Phoxinus phoxinus) from the Pyrenees and the Alps. Biol J Linn Soc 116: 691–703. [CrossRef] [Google Scholar]
- Corral-Lou A, Perea S, Aparicio E, Doadrio I. 2019. Phylogeography and species delineation of the genus Phoxinus Rafinesque, 1820 (Actinopterygii: Leuciscidae) in the Iberian Peninsula. J Zool Syst Evol Res 57: 926–94. [CrossRef] [Google Scholar]
- Cruz A, Llinares C, Martín-Barrio I, Castillo-García G, Arana P, García-Berthou E, Fletcher D, Almeida D. 2022. Comparing morphological, parasitological, and genetic traits of an invasive minnow between intermittent and perennial stream reaches. Freshw Biol 67: 2035–2049. [Google Scholar]
- Cucherousset J, Olden JD. 2011. Ecological impacts of non-native freshwater fishes. Fisheries 36: 215–230. [CrossRef] [Google Scholar]
- Cuinet A, Denys GPJ, Besson S, Marchiset Y, Bouchard J. 2024. First record of the large-scale loach Misgurnus dabryanus (Guichenot, 1872) (Teleostei, Cobitidae) in France. BioInvasions Rec 13: 541–550. [Google Scholar]
- De Santis V, Delmastro GB, Vanetti I, Britton JR, Zaccara S. 2021. Species composition of introduced and natural minnow populations of the Phoxinus cryptic complex in the westernmost part of the Po River Basin (north Italy). Biol Invasions 23: 657–668. [CrossRef] [Google Scholar]
- Degiorigi F, Grandmottet JP, Raymond J-C, Rivier B. 2001. Echantillonnage de l’ichtyofaune lacustre : engins passifs et protocole de prospection. In: Gerdeaux D, (ed.), Gestion Piscicole des Grands Plans d’Eau, Paris: INRA editions, pp 151–182. [Google Scholar]
- Denys GPJ, Dettai A, Persat H, Hautecœur M, Keith P. 2014. Morphological and molecular evidence of three species of pikes Esox spp. (Actinopterygii, Esocidae) in France, including the description of a new species. C R Biol 337: 521–534. [Google Scholar]
- Denys GPJ, Dettai A, Persat H, Daszkiewicz P, Hautecoeur M, Keith P. 2020. Revision of Phoxinus in France with the description of two new species (Teleostei, Leuciscidae). Cybium 44: 205–237. [Google Scholar]
- Denys GPJ, Lestin M, Carré T, Audonnet P, Cazaubon D, Gadaud B, Couturier P, Merilä J, Fusari C-E. 2025. Public aquarium as a key partner for taxonomic studies, an example with French nine-spined sticklebacks Pungitius spp. (Teleostei, Gasterosteiformes). – Cybium, 49: 135–145 . [Google Scholar]
- Esposito A, Denys GPJ, Foata J, Quilichini Y. 2024a. Unclear host taxonomy hinders parasite studies: An up-to-date checklist of the protozoan and metazoan parasites of Phoxinus minnows (Teleostei: Leuciscidae). J Fish Biol 105: 1501–1539. [Google Scholar]
- Esposito A, Denys GPJ, Haÿ V, Godeaux Q, Foata J&, Quilichini Y. 2024b. Multiple introduction pathways of non-native Phoxinus minnows (Teleostei: Leuciscidae) in Corsica revealed by its hidden diversity and their parasites. Biol Invasions 26: 2453–2474. [Google Scholar]
- Forel F-A. 1904. Le Léman, Monographie Limnologique, Vol. 3., Lausanne: E. Rouge, 715 p. [Google Scholar]
- Frost WE. 1943. The natural history of the minnow, Phoxinus phoxinus. J Anim Ecol 12: 139–162. [CrossRef] [Google Scholar]
- Fujii R. 2000. The regulation of motile activity in fish chromatophores. Pigment Cell Res 13: 300–319. [Google Scholar]
- Garcia-Raventós A, Martins FMS, Teixeira A, Sousa R, Froufe E, Varandas S, Lopes-Lima M, Beja P, Filipe AF. 2020. Origin and history of Phoxinus (Cyprinidae) introductions in the Douro Basin (Iberian Peninsula): an update inferred from genetic data. Biol Invasions 22: 2409–2419. [CrossRef] [Google Scholar]
- Geiger MF, Herder F, Monaghan MT, Almada V, Barbieri R, Bariche M, Berrebi P, Bohlen J, Casal‐Lopez M, Delmastro GB, Denys GPJ, Dettai A, Doadrio I, Kalogianni E, Kärst H, Kottelat M, Kovačić M, Laporte M, Lorenzoni M, Marčić Z, Özuluğ M, Perdices A, Perea S, Persat H, Porcelotti S, Puzzi C, Robalo J, Šanda R, Schneider M, Šlechtová V, Stoumboudi M, Walter S, Freyhof J. 2014. Spatial heterogeneity in the Mediterranean Biodiversity Hotspot affects barcoding accuracy of its freshwater fishes. Mol Ecol Resour 14: 1210–1221. [Google Scholar]
- Gozlan R, Britton J, Cowx I, Copp G. 2010. Current knowledge on non-native freshwater fish introductions. J Fish Biol 76(4): 751–786. [CrossRef] [Google Scholar]
- Jurine L. 1825. Histoire abrégée des poissons du lac Léman. Mem Soc Phys Hist Nat Geneve 3: 133–235. [Google Scholar]
- Keith P, Poulet N, Denys G, Changeux T, Persat H. 2020. Les Poissons D'eau Douce de France, 2nd edn. Mèze et Paris: Biotope, Publications scientifiques du Muséum, 704 p. [Google Scholar]
- Kottelat M. 2007. Three new species of Phoxinus from Greece and southern France (Teleostei: Cyprinidae). Ichthyol Explor Freshw 18: 145–162. [Google Scholar]
- Kottelat M, Freyhof J. 2007. Handbook of European Freshwater Fishes. Cornol: Publication Kottelat, 646 p. [Google Scholar]
- Leclercq E, Taylor JF, Migaud H. 2010. Morphological skin colour changes in teleosts. Fish Fish 11: 159–193. [Google Scholar]
- Lorenzoni M, Borghesan F, Carosi A, Ciuffardi L, De Curtis O, Delmastro GB, Di Tizio L, Franzoi P, Maio G, Mojetta A, Nonnis Marzano F, Pizzul E, Rossi G, Scalici M, Tancioni L, Zanetti M. 2019. Check-list dell’ittiofauna delle acque dolci italiane. It J Fresh Ichthyol 5: 239–254. [Google Scholar]
- Lunel G, Lunel A. 1874. Histoire Naturelle des Poissons du Bassin du Léman. Georg, Genève and Lyon, 209 p. [Google Scholar]
- Miró A, Ventura M. 2015. Evidence of exotic trout mediated minnow invasion in Pyrenean high mountain lakes. Biol Inv 17: 791–803. [Google Scholar]
- Miró A, Sabás I, Ventura M. 2018. Large negative effect of non-native trout and minnows on Pyrenean lake amphibians. Biol Conserv 218: 144–153. [CrossRef] [Google Scholar]
- Museth J, Hesthagen T, Sandlund OT, Thorstad EB, Ugedal O. 2007. The history of the minnow Phoxinus phoxinus (L.) in Norway: from harmless species to pest. J Fish Biol 71: 184–195. [CrossRef] [Google Scholar]
- Osorio V, Puig M, Buchaca T, Sabás I, Miró A, Lucati F, Suh J, Pou-Rovira Q, Ventura M. 2022. Non-native minnows cause much larger negative effects than trout on littoral macroinvertebrates of high mountain lakes. Biol Conserv 272: 109637. [Google Scholar]
- Palandačić A, Bravničar J, Zupančič P, Šanda R, Snoj A. 2015. Molecular data suggest a multispecies complex of Phoxinus (Cyprinidae) in the Western Balkan Peninsula. Mol Phylogenet Evol 92: 118–123. [Google Scholar]
- Palandačić A, Naseka A, Ramler D, Ahnelt H. 2017. Contrasting morphology with molecular data: an approach to revision of species complexes based on the example of European Phoxinus (Cyprinidae). BMC Evol Biol 17: 184. [CrossRef] [PubMed] [Google Scholar]
- Palandačić A, Kruckenhauser L, Ahnelt H, Mikschi E. 2020. European minnows through time: museum collections aid genetic assessment of species introductions in freshwater fishes (Cyprinidae: Phoxinus species complex). Heredity 124: 412–422. [Google Scholar]
- Palandačić A, Witman K, Spikmans F. 2022. Molecular analysis reveals multiple native and alien Phoxinus species (Leusciscidae) in the Netherlands and Belgium. Biol Invasions 24: 2273–2283. [CrossRef] [Google Scholar]
- Palandačić A, Diripasko O, Kirchner S, Stefanov T, Bogutskaya N. 2024a. An integrative approach highlights the discrepancy in the genetic, phenotypic, and presumptive taxonomic structure of Phoxinus (Actinopterygii, Leuciscidae, Phoxininae) in Bulgaria. J Fish Biol 105: 214–238. [Google Scholar]
- Palandačić A, Chai MJ, Bogutskaya N, Reier S, Friedrich R, Wanzenböck S, Wanzenböck J, Rund H, Glaser F, Mikschi E. 2024b. Then and now: citizen scientists help assess the changing biodiversity of minnows in Austria. ARPHA Proceedings 1: 31–36. [Google Scholar]
- Pedroli J-C, Zaugg B, Kirchhofer A. 1991. Verbreitungsatlas der Fische und Rundmäuler der Schweiz / Atlas de Distribution des Poissons et Cyclostomes de Suisse. Neuchâtel: Schweizerisches Zentrum für die kartografische Erfassung der Fauna, 207 p. [Google Scholar]
- Persat H, Keith P, Denys GPJ. 2020. Biogéographie et historique de la mie en place des peuplements ichtyologiques de France métropolitaines. In: Keith P, Poulet N, Denys G, Changeux T, Persat H, (eds.), Les Poissons D’eau Douce de France, 2nd edn. Mèze, Paris: Biotope, Publications scientifiques du Muséum, pp. 43–97. [Google Scholar]
- Ramler D, Palandačić A, Delmastro GB, Wanzenböck J, Ahnelt H. 2017. Morphological divergence of lake and stream Phoxinus of Northern Italy and the Danube basin based on geometric morphometric analysis. Ecol Evol 7: 572–584. [CrossRef] [PubMed] [Google Scholar]
- Raymond J-C, Bolard A, Persat H. 2010. Premier signalement de Salaria fluviatilis dans le Lac Léman. Cybium 34: 401–402. [Google Scholar]
- Reier S, Bogutskaya N, Palandačić A. 2022. Comparative phylogeography of Phoxinus, Delminichthys, Phoxinellus and Telestes in dinaric karst: which factors have influenced their current distributions? Diversity 14: 526. [CrossRef] [Google Scholar]
- Reier S, Trontelj P, Kruckenhauser L, Kapun M, Wanka A, Palandačić A. 2025. Factors beyond karstification have shaped the population structure of a surface-dwelling minnow (Phoxinus lumaireul) able to disperse underground. Evol Appl 18: e70104. [Google Scholar]
- Ribeiro F, Leunda P. 2012. Non-native fish impacts on Mediterranean freshwater ecosystems: current knowledge and research needs. Fisheries Manag Ecol 19(2): 142–156. [Google Scholar]
- Scharnweber K. 2020. Morphological and trophic divergence of lake and stream minnows (Phoxinus phoxinus). Ecol Evol 10: 8358–8367. [CrossRef] [PubMed] [Google Scholar]
- Splendiani A, Berrebi P, Tougard C, Righi T, Reynaud N, Fioravanti T, Lo Conte P, Delmastro GB, Baltieri M, Ciuffardi L, Candiotto A, Sabatini A, Caputo Barucchi V. 2020. The role of the south-western Alps as unidirectional corridor for Mediterranean brown trout (Salmo trutta complex) lineages. Biol J Linn Soc 131: 909–926. [Google Scholar]
- Sternberg N, Bodenheim A, Oriowo TO, Podsiadlowski L, Stange M. 2025. Human impacts on the distribution and genetic diversity of Eurasian minnows (Phoxinus: Leuciscidae) in the Rhenish Massif. Knowl Manag Aquat Ecosyst 426: 20. [Google Scholar]
- Takács P, Vitál Z, Ferincz Á, Staszny Á. 2016. Repeatability, reproducibility, separative power and subjectivity of different fish morphometric analysis methods. PLOS ONE 11: e0157890. [Google Scholar]
- TAXREF. 2025. TAXREF v18.0, référentiel taxonomique pour la France. PatriNat (OFB-CNRS-MNHN-IRD), Muséum national d'Histoire naturelle, Paris. https://inpn.mnhn.fr/telechargement/referentielEspece/taxref/18.0/menu [Google Scholar]
- Thaulow J, Borgstrøm R, Heun M. 2013. Genetic analyses of European minnow, Phoxinus phoxinus, in a river system outside its native range indicate multiple invasions from different sources. Fish Manag Ecol 21: 75–81. [Google Scholar]
- Tiberti R, Buchaca T, Cruset Tonietti E, Iacobelli L, Maini M, Ribelli F, Pou Rovira Q, Ventura M. 2022. Minnow introductions in mountain lakes result in lower salmonid densities. Biol Invasions 24: 2285–2289. [CrossRef] [Google Scholar]
- Turan D, Bayçelebi E, Özuluğ M, Gaygusuz Ö, Aksu İ. 2023. Phoxinus abanticus, a new species from the Lake Abant drainage in Türkiye (Teleostei: Leuciscidae). J Fish Biol 102: 1157–1167. [CrossRef] [PubMed] [Google Scholar]
- Vissio P, Darias M, Di Yorio M, Pérez Sirkin D, Delgadin T. 2021. Fish skin pigmentation in aquaculture: the influence of rearing conditions and its neuroendocrine regulation. Gen Comp Endocrinol 301: 113662. [Google Scholar]
- Vucić M, Jelić D, Žutinić P, Grandjean F, Jelić M. 2018. Distribution of Eurasian minnows (Phoxinus: Cypriniformes) in the Western Balkans. Knowl Manag Aqu Ecosyst 419: 11. [Google Scholar]
- Vucić M, Jelić M, Klobučar G, Jelić D, Gan HM, Austin C, Guyonnet D, Giraud I, Becking T, Grandjean F. 2022. A new set of microsatellite markers for Phoxinus lumaireul senso lato, Phoxinus marsilii and Phoxinus krkae for population and molecular taxonomic studies. J Fish Biol 101: 1225–1234. [Google Scholar]
- Witkowski A, Rogowska M. 1992. Breeding tubercles in some European cyprinid fishes (Osteichthyes, Cypriniformes: Cyprinidae). Zool Abh Dresden 46: 131–156. [Google Scholar]
Cite this article as: Denys G, Raymond J-C, Delmastro G. 2026. Characterisation of the nuptial colouration pattern of Phoxinus lumaireu Schinz, 1840 (Teleostei, Leuciscidae) and first record in France. Int. J. Lim. 62: 1. https://doi.org/10.1051/limn/2025012
All Tables
Origin of Italian minnows genotyped as P. lumaireul according to previous studies, and used for the characterisation of the nuptial colouration pattern. Museums abbrevations: Museo Civico di Storia Naturale di Carmagnola, Carmagnola, Italy (MCCI); Muséum national d’Histoire naturelle, Paris, France (MNHN); Naturhistorisches Museum, Zoologische Abteilung, Fische, Vienna, Austria (NMW); Zoologisches Forschungsmuseum Alexander König, Bonn, Germany (ZFMK).
Morphological observations and approximated lengths ratios taken on photograph (Fig. 4; in pixel numbers) of the male minnow caught in the Lake Geneva the 31st May 2010 compared to data from Kottelat and Freyhof (2007) (*), Palandačić et al. (2017) ($) and Denys et al. (2020) (£) on P. csikii, P. lumaireul and P. septimaniae from morphomeristic analyses with measurements issued from digital calliper; diagnostic characters are in bold; for the snout shape see the Supplementary data 2; HL means head length.
All Figures
 |
Fig. 1 Sampling locations (red squares) of minnows in the Po catchment in the Italian Piedmont (green box) where specimens of P. lumaireul were caught and used for the nuptial colouration characterisation (see Tab. 1, Figs. 2, 4b, and 5c), as well as in the French part of the Lake Geneva (red box) where the minnow specimen of unknown species was photographed and presented in Fig. 4a. Locations: 1) Varaita Torrent at Costigliole Saluzzo 44°33’49’’N – 7°28’29’’E, 2) Chisone Torrent at Pinerolo 45°51’42’’N – 7°20’41’’E, 3) Lago Laus, Roreto Chisone 45°03’00’’N – 7°08’08’’E, 4) Sangone Torrent at Trana 45°01’43’’N – 7°23’42’’E, 5) Lake Geneva at Publier 46°23’46”N – 6°32’22”E. |
| In the text | |
 |
Fig. 2 Phoxinus lumaireul in the Po catchment with alive nuptial colourations: male (68 mm TL) from the Varaita Torrent at Costigliole Saluzzo (a), five males (57 to 64 mm TL) from the Chisone Torrent at Pinerolo (b), male (not measured) from the Lago del Lau at Roreto Chisone (c), two females (75 mm TL (d) and 81 mm TL (e)) from the Sangone Torrent at Trana, nuptial colouration pattern for male (f) and female (g); photo credits: G. Delmastro. |
| In the text | |
 |
Fig. 3 The five zones on the minnow’s flank characterising the nuptial colouration pattern (a) following Denys et al. (2020), and both male and female nuptial colouration patterns for respectively Phoxinus csikii (b) and Phoxinus septimaniae (c) according to Denys et al. (2020); drawings adapted and modified from Denys et al. (2020). |
| In the text | |
 |
Fig. 4 Male minnow (65 mm TL, a) in nuptial colouration with an unknown species origin caught in the Lake Geneva at Publier the 31st May 2010 and one male Phoxinus lumaireul (83 mm TL, b; identified by Geiger et al. (2014)) from the Sangone Torrent at Trana the 15th June 2011; photo credits: respectively J.-C. Raymond / OFB and G. Delmastro. |
| In the text | |
 |
Fig. 5 Minnows drawings (a,b; modified symmetrically to the left side) from Lunel and Lunel (1874): a male identified as Phoxinus csikii by Denys et al. (2020) (a), another male having the nuptial coulouration pattern of Phoxinus lumaireul (b) like the male (57 mm TL) from the Chisone Torrent at Pinerolo (c; photo credits: G. Delmastro). |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


